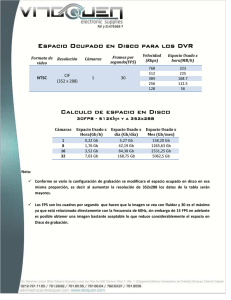
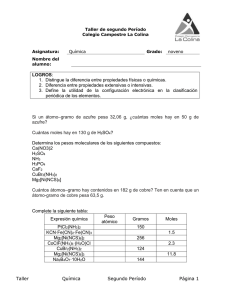
What is a Chemical Engineer?
A chemical engineer is a professional man experienced in the design, construction, and operation
of plants in which materials undergo chemical or physical change.
British Institution of Chemical Engineers, Inaugural Meeting, 1922
We have come to the conclusion that a chemical engineer as such does not in reality exist.
A. Duckham, Presidential Address, British Institution of Chemical Engineers, 1924
Abstract
Chemical absorption of H2S, NH3 and HCl from syngas derived from the gasification of SRF using
caustic solutions is studied. These inorganic pollutants are corrosive and harmful to the environment,
making their elimination necessary. Water and NaOH caustic solution mixed with NaOCl were
investigated as scrubbing liqueur. Several models using Aspen Plus, HYSYS and homemade routines
(solved with EES) were developed. The individual models for H2S, NH3 and HCl removal were validated
by comparison with literature experimental data. Some configurations were simulated to understand their
effectiveness for cleaning the gas for electricity generation through burning in gas boilers and engines
(targets 80 mg/Nm3 of H2S, 50 mg/Nm3 of NH3 and 50 mg/Nm3 of HCl). A preliminary economic
analysis was made by comparing the proposed design based on NaOH with traditional absorption with
amines (MDEA). The present study does not consider the absorption of CO2. Although, the use of NaOH
is not appropriate for gas with high CO2/H2S and standard scrubbing contact times (as syngas from waste
in packed towers where the selectivity to H2S is very limited), the present work establishes the
methodology towards processes with higher selectivity (short-contact time scrubbing process and/or the
use of more selective scrubbing solvents).
Resumen
La absorción química de H2S, NH3 y HCl de una corriente de syngas derivada de la gasificación de SRF
utilizando cáusticos es estudiada. Estos contaminantes inorgánicos son corrosivos y dañinos para el medio
ambiente por lo que es necesaria su eliminación. Una solución cáustica de NaOH y agua más NaOCl
como oxidante se ha investigado como líquido absorbente. Varios modelos se han desarrollado utilizando
Aspen Plus, Hysys y rutinas realizadas en EES. Los modelos individuales para la eliminación de H 2S,
NH3 y HCl fueron validados comparándolos con datos experimentales. Algunas configuraciones fueron
simuladas para comprender la eficiencia del proceso de absorción para cumplir los requisitos de limpieza
de los equipos de generación eléctrica en motores de combustión (límites de 80 mg/Nm3 de H2S, 50
mg/Nm3 de NH3 y 50 mg/Nm3 de HCl). Finalmente, se ha realizado un análisis económico preliminar
comparando el presente diseño basado en NaOH frente a las alternativas tradicionales regenerativas como
el de MDEA. Nótese, que el presente estudio no tiene en cuenta la absorción de CO2. Es importante este
factor, ya que el NaOH no es el absorbente apropiado para gases con una alta relación CO2/H2S en los
tiempos de contacto estándar (ya que la selectividad a H2S en torres de relleno está muy limitada). El
presente trabajo establece una metodología para futuras investigaciones hacia procesos de una alta
selectividad (procesos de corto tiempo de contacto o procesos con absorbentes más selectivos).