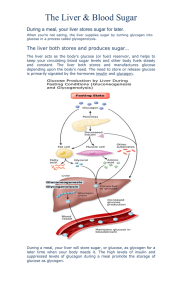
15/9/21 20:09 https://emedicine.medscape.com/article/122122-print emedicine.medscape.com Hypoglycemia Updated: Aug 23, 2021 Author: Osama Hamdy, MD, PhD; Chief Editor: Romesh Khardori, MD, PhD, FACP Overview Practice Essentials Hypoglycemia is characterized by a reduction in plasma glucose concentration to a level that may induce symptoms or signs such as altered mental status and/or sympathetic nervous system stimulation. This condition typically arises from abnormalities in the mechanisms involved in glucose homeostasis. The most common cause of hypoglycemia in patients with diabetes is injecting a shot of insulin and skipping a meal or overdosing insulin.[1] The image below depicts a diagnostic algorithm for hypoglycemia. Diagnostic algorithm. A systematic approach is often required to establish the true cause of hypoglycemia, using an algorithmic approach. Signs and symptoms of hypoglycemia The glucose level at which an individual becomes symptomatic is highly variable (threshold generally at < 50 mg/dL). Carefully review the patient's medication and drug history for potential causes of hypoglycemia (eg, new medications, insulin usage or ingestion of an oral hypoglycemic agent, possible toxic ingestion). The patient’s medical and/or social history may reveal the following: Diabetes mellitus, renal insufficiency/failure, alcoholism, hepatic cirrhosis/failure, other endocrine diseases, or recent surgery Central nervous system: Headache, confusion, personality changes Ethanol intake and nutritional deficiency https://emedicine.medscape.com/article/122122-print 1/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Weight reduction, nausea and vomiting Fatigue, somnolence Neurogenic or neuroglycopenic symptoms of hypoglycemia may be categorized as follows: Neurogenic (adrenergic) (sympathoadrenal activation) symptoms: Sweating, shakiness, tachycardia, anxiety, and a sensation of hunger Neuroglycopenic symptoms: Weakness, tiredness, or dizziness; inappropriate behavior (sometimes mistaken for inebriation), difficulty with concentration; confusion; blurred vision; and, in extreme cases, coma and death Reactive hypoglycemia includes the following features: More common in overweight and obese people who are insulin-resistant May be a frequent precursor to type 2 diabetes Possible higher risk in patients with a family history of type 2 diabetes or insulin-resistance syndrome True loss of consciousness is highly suggestive of an etiology other than reactive hypoglycemia. Gestational hypoglycemia may have the following features[2] : More frequent in women younger than 25 years More frequent in women with a preexisting medical condition Less frequent in women whose prepregnancy body mass index was ≥30 kg/m2 Greater risk of preeclampsia/eclampsia in affected women See Clinical Presentation for more detail. Diagnosis of hypoglycemia Rapid diagnosis and treatment is essential in any patient with suspected hypoglycemia, regardless of the cause. The Whipple triad is characteristically present: documentation of low blood sugar, presence of symptoms, and reversal of these symptoms when the blood glucose level is restored to normal. Physical findings, however, are nonspecific in hypoglycemia and are generally related to the central and autonomic nervous systems. Examination includes the following: Vital signs Head, eyes, ears, nose, and throat Cardiovascular Neurologic Pulmonary Gastrointestinal Dermatologic Elderly persons exhibit fewer symptoms of hypoglycemia, and their threshold of plasma glucose is lower at presentation than in younger persons. Laboratory studies Patients with no previous history of hypoglycemia require a complete workup to find a potentially treatable disease. Laboratory studies that should be obtained include the following: Glucose and electrolyte levels (including calcium, magnesium) Oral glucose tolerance test and/or 72-hour fasting plasma glucose https://emedicine.medscape.com/article/122122-print 2/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Complete blood count Other tests that may be helpful including the following: Blood cultures Urinalysis Serum insulin, cortisol levels, and thyroid hormone levels C-peptide levels Proinsulin Imaging studies Imaging modalities to evaluate insulinomas may include the following: CT scanning MRI Octreotide scanning Procedures Selective percutaneous transhepatic venous sampling may be performed to localize an insulinoma to the pancreatic head, body, or tail. Selective arteriography is also often helpful in localizing insulin-secreting lesions. See Workup for more detail. Management of hypoglycemia Pharmacotherapy The mainstay of therapy for hypoglycemia is glucose. Other medications may be administered based on the underlying cause or the accompanying symptoms. Medications used in the treatment of hypoglycemia include the following: Glucose supplements (eg, dextrose) Glucose-elevating agents (eg, glucagon, glucagon intranasal) Inhibitors of insulin secretion (eg, diazoxide, octreotide) Antineoplastic agents (eg, streptozocin) Other therapies Fasting hypoglycemia: Dietary therapy (frequent meals/snacks preferred, especially at night, with complex carbohydrates); IV glucose infusion; IV octreotide Reactive hypoglycemia: Dietary therapy (restriction of refined carbohydrates, avoidance of simple sugars, increased meal frequency, increased protein and fiber); alpha-glucosidase inhibitors Surgery Definitive treatment for fasting hypoglycemia caused by a tumor is surgical resection. The success rate is good for benign isletcell adenomas, and the success rate for malignant islet-cell tumors can be as high as 50%. See Treatment and Medication for more detail. Background https://emedicine.medscape.com/article/122122-print 3/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Hypoglycemia is a clinical situation characterized by a reduction in plasma glucose concentration to a level that may induce symptoms or signs such as altered mental status and/or sympathetic nervous system stimulation. The glucose level at which an individual becomes symptomatic is highly variable, although a plasma glucose level less than 50 mg/dL is generally considered the threshold. Hypoglycemia typically arises from abnormalities in the mechanisms involved in glucose homeostasis. To diagnose hypoglycemia, the Whipple triad is characteristically present. This triad includes the documentation of low blood sugar, presence of symptoms, and reversal of these symptoms when the blood sugar level is restored to normal. See a diagnostic algorithm for hypoglycemia, below. Diagnostic algorithm. A systematic approach is often required to establish the true cause of hypoglycemia, using an algorithmic approach. See also Pediatric Hypoglycemia, Neonatal Hypoglycemia, Emergent Management of Acute Symptoms of Hypoglycemia, and Congenital Hyperinsulinism. Pathophysiology Hypoglycemic symptoms are related to sympathetic activation and brain dysfunction secondary to decreased levels of glucose. Stimulation of the sympathoadrenal nervous system leads to sweating, palpitations, tremulousness, anxiety, and hunger. Reduction in cerebral glucose availability (ie, neuroglycopenia) can manifest as confusion, difficulty with concentration, irritability, hallucinations, focal impairments (eg, hemiplegia), and, eventually, coma and death. The adrenergic symptoms often precede the neuroglycopenic symptoms and, thus, provide an early warning system for the patient. Studies have shown that the primary stimulus for the release of catecholamines is the absolute level of plasma glucose; the rate of decrease of glucose is less important. Previous blood sugar levels can influence an individual's response to a particular level of blood sugar. However, it is important to note that a patient with repeated hypoglycemia can have almost no symptoms (hypoglycemic unawareness). The threshold at which a patient feels the hypoglycemic symptoms decreases with repeated episodes of hypoglycemia. A study by Zhong et al indicated that in patients with type 2 diabetes mellitus, a proximal hemoglobin A1c (HbA1c) level above or below the reference level of 7.0% increases the risk of a first incidence of hypoglycemia requiring hospitalization (HH). The investigators found that when the proximal HbA1c level ranges between 4.0% and 6.5%, every 0.5% level increase reduces the first HH risk, while for patients with a proximal HbA1c level of 8.0-11.5%, the risk of first HH rises with each 0.5% increase in HbA1c. The investigators also found that current sulfonylurea users in the study did not exhibit this U-shaped phenomenon but that current insulin users did.[3] Etiology https://emedicine.medscape.com/article/122122-print 4/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Causes of hypoglycemia are varied, but it is seen most often in diabetic patients. Hypoglycemia may result from medication changes or overdoses, infection, diet changes, metabolic changes over time, or activity changes; however, no acute cause may be found. Other causes include alimentary problems, idiopathic causes, fasting, insulinoma, endocrine problems, extrapancreatic causes, hepatic disease, and bariatric surgery, along with additional miscellaneous etiologies. In a multicenter, retrospective cohort study, Riegger et al reported that independent risk factors for intraoperative hypoglycemia in children include age under 5 years, weight for age below the fifth percentile, American Society of Anesthesiologists status of III or above, the presence of a gastric or jejunal tube, poor feeding, and abdominal surgery. Eighty percent of hypoglycemia cases were in children under age 5 years and in youngsters weighing under 20 kg.[4] A study by van Furth et al indicated that both dumping syndrome and post–bariatric surgery hypoglycemia can be attributed to a common etiology, with glucagon-like peptide 1 (GLP-1) and peptide YY (PYY) being key to the development of each. The investigators point out that L-cells, which secrete GLP-1 and PYY, are affected by bariatric surgery, with, for example, an increase in L-cells in the perianastomotic jejunum occurring after Roux-en-Y gastric bypass.[5] Fasting hypoglycemia Nesidioblastosis is a rare cause of fasting hypoglycemia in infants and an extremely rare cause in adults. This condition is characterized by a diffuse budding of insulin-secreting cells from pancreatic duct epithelium and pancreatic microadenomas of such cells. Causes of fasting hypoglycemia usually diagnosed in infancy or childhood include inherited liver enzyme deficiencies that restrict hepatic glucose release (deficiencies of glucose-6-phosphatase, fructose-1,6-diphosphatase, phosphorylase, pyruvate carboxylase, phosphoenolpyruvate carboxykinase, or glycogen synthetase). Inherited defects in fatty acid oxidation, including those resulting from systemic carnitine deficiency and inherited defects in ketogenesis (3-hydroxy-3-methylglutaryl-CoA lyase deficiency), cause fasting hypoglycemia by restricting the extent to which nonneural tissues can derive their energy from plasma free fatty acids (FFA) and ketones during fasting or exercise. This results in an abnormally high rate of glucose uptake by nonneural tissues under these conditions. Several cases of nesidioblastosis were reported recently after gastric bypass surgery. Drugs Ethanol (including propranolol plus ethanol), haloperidol, pentamidine, quinine, salicylates, and sulfonamides ("sulfa drugs") have been associated with hypoglycemia. Other drugs that may be related to this condition include oral hypoglycemics, phenylbutazone, insulin, bishydroxycoumarin, p-aminobenzoic acid, propoxyphene, stanozolol, hypoglycin, carbamate insecticide, disopyramide, isoniazid, methanol, methotrexate, tricyclic antidepressants, cytotoxic agents, organophosphates, didanosine, chlorpromazine, fluoxetine, sertraline, fenfluramine, trimethoprim, 6-mercaptopurine, thiazide diuretics, thioglycolate, tremetol, ritodrine, disodium ethylenediaminetetraacetic acid (EDTA), clofibrate, angiotensin converting enzyme (ACE) inhibitors, and lithium. A study by Fournier and colleagues indicates that treatment for pain with the opioid analgesic tramadol increases a patient’s risk of being hospitalized for hypoglycemia. Information from the United Kingdom Clinical Practice Research Datalink and the Hospital Episode Statistics database was analyzed for 28,110 patients who were newly prescribed tramadol and 305,924 individuals who were newly prescribed codeine, all for noncancer pain, with 11,019 controls also included in the study. Using case-control, cohort, and case-crossover analysis, the investigators found that tramadol increased the risk of hospitalization for hypoglycemia by more than three-fold, with the risk particularly elevated in the first 30 days of treatment. The actual risk was small, however, occurring in about 7 patients per 10,000 annually.[6, 7] A study by Eriksson et al indicated that in patients with type 2 diabetes undergoing second-line treatment, the combination of metformin and sulfonylurea carries a greater risk for severe hypoglycemia, cardiovascular disease, and all-cause mortality than does the combination of metformin and dipeptidyl peptidase-4 inhibitor (DPP4i).[8] Similarly, a study by Gautier et al found that patients with type 2 diabetes treated with metformin plus insulin secretagogues (such as sulfonylurea or glinide) were more likely to experience hypoglycemia than were those treated with metformin plus DPP4i while starting insulin. Both groups achieved similar glycemic control.[9] Surreptitious sulfonylurea use/abuse Factitious, or self-induced, hypoglycemia can be seen in healthcare workers or in relatives who care for diabetic family members at home.[10] (See Type 1 Diabetes Mellitus and Type 2 Diabetes Mellitus for further discussion, including the diagnostic use of C-peptide levels and HbA1c.) Exogenous insulin https://emedicine.medscape.com/article/122122-print 5/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Surreptitious use of insulin may be seen, typically among those likely to have access to insulin. Measurement of insulin level along with C-peptide is very crucial in making this diagnosis. Endogenous insulin or insulin-receptor–mediated hypoglycemia Sources of endogenous insulin include insulin-producing tumors of pancreas and non–beta-cell tumors. Insulin-producing tumors of pancreas Islet cell adenoma or carcinoma (insulinoma) is an uncommon and usually curable cause of fasting hypoglycemia and is most often diagnosed in adults. It may occur as an isolated abnormality or as a component of the multiple endocrine neoplasia type I (MEN I) syndrome. Carcinomas account for only 10% of insulin-secreting islet cell tumors. Hypoglycemia in patients with islet cell adenomas results from uncontrolled insulin secretion, which may be clinically determined during fasting and exercise. Approximately 60% of patients with insulinoma are female. Insulinomas are uncommon in persons younger than 20 years and are rare in those younger than 5 years. The median age at diagnosis is about 50 years, except in patients with MEN syndrome, in which the median age is in the mid third decade of life. Ten percent of patients with insulinoma are older than 70 years. Non–beta-cell tumors Hypoglycemia may also be caused by large non–insulin-secreting tumors, most commonly retroperitoneal or mediastinal malignant mesenchymal tumors. The tumor secretes abnormal insulinlike growth factor (large IGF-II), which does not bind to its plasma binding proteins. This increase in free IGF-II exerts hypoglycemia through the IGF-I or the insulin receptors. The hypoglycemia is corrected when the tumor is completely or partially removed and usually recurs when the tumor regrows. Reactive hypoglycemia Reactive hypoglycemia can be idiopathic, due to alimentary problems, or a result of congenital enzyme deficiencies. Alimentary hypoglycemia is another form of reactive hypoglycemia that occurs in patients who have had previous upper gastrointestinal (GI) surgical procedures (gastrectomy, gastrojejunostomy, vagotomy, pyloroplasty) and allows rapid glucose entry and absorption in the intestine, provoking excessive insulin response to a meal. This may occur within 1-3 hours after a meal. Very rare cases of idiopathic alimentary hypoglycemia occur in patients who have not had GI operations. Congenital enzyme deficiencies include hereditary fructose intolerance, galactosemia, and leucine sensitivity of childhood. In hereditary fructose intolerance and galactosemia, an inherited deficiency of a hepatic enzyme causes acute inhibition of hepatic glucose output when fructose or galactose is ingested. Leucine provokes an exaggerated insulin secretory response to a meal and reactive hypoglycemia in patients with leucine sensitivity of childhood. Other causes of hypoglycemia include the following, singly or in combination (eg, chronic renal failure and sulfonylurea ingestion): Autoimmune hypoglycemia: Insulin antibodies and insulin receptor antibodies Hormonal deficiencies: Hypoadrenalism (cortisol), hypopituitarism (growth hormone) (in children), glucagons deficiency (rare), and epinephrine (very rare) Critical illnesses: Cardiac, hepatic, and renal diseases; sepsis with multiorgan failure Exercise (in patients with diabetes treated with diabetes medications) Pregnancy Renal glycosuria Ketotic hypoglycemia of childhood Adrenal insufficiency Hypopituitarism Starvation Artifact https://emedicine.medscape.com/article/122122-print 6/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Epidemiology The incidence of hypoglycemia in a population is difficult to ascertain. Patients and physicians frequently attribute symptoms (eg, anxiety, irritability, hunger) to hypoglycemia without documenting the presence of low blood sugar. The true prevalence of hypoglycemia, with blood sugar levels below 50 mg/dL, is generally 5-10% of people presenting with symptoms suggestive of hypoglycemia. A Brazilian study, by Lamounier et al, found that during a 4-week prospective evaluation period, at least one hypoglycemic event occurred in 91.7% of study patients with type 1 diabetes and in 61.8% of those with type 2 diabetes. This included nocturnal hypoglycemia in 54.0% and 27.4% of patients, respectively; asymptomatic hypoglycemia in 20.6% and 10.6% of patients, respectively; and severe hypoglycemic events in 20.0% and 10.3% of patients, respectively.[11] Hypoglycemia is a known complication of several medications, and the incidence is difficult to determine with any certainty. In addition, this condition is a known complication of many therapies for diabetes; therefore, the incidence of hypoglycemia in a population of people with diabetes is very different from that in a population of people without diabetes.[12, 13, 14, 15, 16, 17] Insulin-producing tumors are a rare but important treatable cause of hypoglycemia, with an annual US incidence of 1-2 cases per million persons per year. Reactive hypoglycemia is reported most frequently by women aged 25-35 years; however, other causes of hypoglycemia are not associated with a sex predilection. The average age of a patient diagnosed with an insulinoma is the early 40s, but cases have been reported in patients ranging from birth to age 80 years.[18] Prognosis The prognosis of hypoglycemia depends on the cause of this condition, its severity, and its duration. If the cause of fasting hypoglycemia is identified and treated early, the prognosis is excellent. If the problem is not curable, such as an inoperable malignant tumor, the long-term prognosis is poor. However, note that these tumors may progress rather slowly. Severe and prolonged hypoglycemia can be life threatening and may be associated with increased mortality in patients with diabetes. If the patient has reactive hypoglycemia, symptoms often spontaneously improve over time, and the long-term prognosis is very good. Reactive hypoglycemia is often treated successfully with dietary changes and is associated with minimal morbidity. Mortality is not observed. Untreated reactive hypoglycemia may cause significant discomfort to the patient, but long-term sequelae are not likely. A study by Boucai et al found that drug-associated hypoglycemia was not associated with increased mortality risk among patients admitted to general wards. This suggests that hypoglycemia may be a marker of disease burden and not a direct cause of death.[19] Presentation History The patient's medication and drug history should be reviewed carefully for potential causes of hypoglycemia. Inquire if the patient is taking any new medications. A history of insulin usage or ingestion of an oral hypoglycemic agent may be known, and possible toxic ingestion should be considered. Injecting a shot of insulin and skipping a meal or overdosing insulin is the most common cause in patients with diabetes. It is important to determine how the patient is taking oral insulin secretagogues, particularly sulfonylureas. Often, those with nocturnal hypoglycemia are taking the drug late at night after supper. A careful history will uncover this. The medical history may include diabetes mellitus, renal insufficiency/failure, alcoholism, hepatic cirrhosis/failure, other endocrine diseases, or recent surgery. However, obtaining an accurate medical history may be difficult if the patient's mental status is altered. Central nervous system (CNS) symptoms include headache, confusion, and personality changes. The social history may include ethanol intake and nutritional deficiency. Review systems for weight reduction, fatigue, somnolence, nausea and vomiting, and headache. Look for other symptoms suggesting infection. https://emedicine.medscape.com/article/122122-print 7/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Neurogenic or neuroglycopenic symptoms Symptoms of hypoglycemia may be categorized as neurogenic (adrenergic) or neuroglycopenic. Sympathoadrenal activation symptoms include sweating, shakiness, tachycardia, anxiety, and a sensation of hunger. Neuroglycopenic symptoms include weakness, tiredness, or dizziness; inappropriate behavior (sometimes mistaken for inebriation), difficulty with concentration; confusion; blurred vision; and, in extreme cases, coma and death. The timing of onset of symptoms relative to the time of meal ingestion is crucial in the evaluation of a patient with hypoglycemia. Fasting hypoglycemia typically occurs in the morning before eating or during the day, particularly in the afternoon if meals are missed or delayed. Postprandial hyperglycemia typically occurs 2-4 hours after eating food, especially when meals contain high levels of simple carbohydrates. Postprandial symptoms are typically due to reactive causes, but some patients with insulinoma also may present with postprandial symptoms. About 4-6 hours after food ingestion, plasma glucose concentrations are 80-90 mg/dL, and rates of glucose utilization and production are approximately 2 mg/kg/min. Glucose production is primarily (70-80%) from hepatic glycogenolysis, with a lesser contribution (20-25%) from hepatic gluconeogenesis. A study by Feil et al found a high risk of hypoglycemia among patients with dementia and cognitive impairment.[20] Reactive hypoglycemic symptoms Reactive hypoglycemia seldom causes glucose levels to drop low enough to induce severe neuroglycopenic symptoms; therefore, a history of true loss of consciousness is highly suggestive of an etiology other than reactive hypoglycemia. Reactive hypoglycemia has been suggested to be more common in overweight and obese people who are insulin-resistant, and it may be a frequent precursor to type 2 diabetes. Therefore, patients who have a family history of type 2 diabetes or insulinresistance syndrome (ie, hypertension, hyperlipidemia, obesity) may be at higher risk for developing hypoglycemia. Gestational hypoglycemia In a study of maternal hypoglycemia, Pugh et al found that hypoglycemia occurred more frequently in women younger than 25 years and those who had a preexisting medical condition.[2] Hypoglycemia was less frequent in women whose prepregnancy body mass index (BMI) was ≥ 30 kg/m2. The investigators also found that patients with hypoglycemia were at greater risk of preeclampsia/eclampsia. Physical Examination Physical findings are nonspecific in hypoglycemia and generally are related to the central and autonomic nervous systems. Assess vital signs for hypothermia, tachypnea, tachycardia, hypertension, and bradycardia (neonates). The head, eyes, ears, nose, and throat (HEENT) examination may indicate blurred vision, pupils normal to fixed and dilated, icterus (usually cholestatic due to hepatic disease), and parotid pain (due to endocrine causes). Cardiovascular disturbances may include tachycardia (bradycardia in neonates), hypertension or hypotension, and dysrhythmias. Neurologic conditions include coma, confusion, fatigue, loss of coordination, combative or agitated disposition, stroke syndrome, tremors, convulsions, and diplopia. Respiratory disturbances may include dyspnea, tachypnea, and acute pulmonary edema. Gastrointestinal disturbances may include nausea and vomiting, dyspepsia, and abdominal cramping. The patient's skin may be diaphoretic and warm or show signs of dehydration with decrease in turgor. Symptoms of hypoglycemia are fewer in elderly persons and they frequently appear at a lower threshold of plasma glucose than in younger persons. DDx Diagnostic Considerations https://emedicine.medscape.com/article/122122-print 8/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Because the consequences of hypoglycemia can be devastating and an antidote is readily available, diagnosis and treatment must be rapid in any patient with suspected hypoglycemia, regardless of the cause. Patients with no previous history of hypoglycemia require a complete workup to find a potentially treatable disease. Careful consideration should be given to all diabetic patients presenting with hypoglycemia. New medications, activity changes, and infection should be considered. Early in the course of non–insulin-dependent diabetes, patients may experience episodes of hypoglycemia several hours after meals. The symptoms generally are brief and respond spontaneously. Conditions such as Jamaican vomiting sickness, ingestion of ethanol-containing mouthwash or cologne (children), gastric surgery, potassium administration during periodic attacks of paralysis, excessive muscular activity, diarrhea (childhood) can also cause hypoglycemia. The following should also be considered when evaluating a patient with hypoglycemia: Hepatic disease: (eg, hepatic failure, cirrhosis, galactose intolerance, fructose intolerance, glycogen storage diseases) [21] Transient ischemic attacks Cardiac dysrhythmia Endocrine disorders (eg, pheochromocytoma, Addison disease, glucagon deficiency, carcinomas, extrahepatic tumors) Substance abuse (eg, cocaine, ethanol, salicylates, beta-blockers, pentamidine) Hypoglycemic agents (eg, insulin, oral hypoglycemic agents) Nutritional disorders (eg, prolonged starvation before anesthesia, protein calorie malnutrition, L-leucine-sensitive hypoglycemic defect in children, low-calorie ketogenic diet, renal disease) Autoimmune disorders (eg, Graves disease) Central nervous system (CNS) disorders Psychogenic Differential Diagnoses Addison Disease Adrenal Crisis Alcoholism Anxiety Disorders Cardiogenic Shock Hypopituitarism (Panhypopituitarism) Insulinoma Pseudohypoglycemia Workup Workup Approach Considerations Search for a source of infection. Studies should be considered to rule out the possibility of a concurrent occult infection contributing to the new hypoglycemic episode (eg, complete physical examination, chest radiography (particularly in diabetic patients presenting with hypoglycemia), urinalysis, blood cultures). https://emedicine.medscape.com/article/122122-print 9/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Check liver function tests, serum insulin levels, and cortisol and thyroid levels. Proinsulin normally represents less than 20% of total immunoreactive insulin; in patients with islet-cell tumors, proinsulin may contribute as much as 70% of insulin immunoreactivity. Provocative tests involving the administration of arginine, leucine, calcium, glucagon, or tolbutamide are generally of limited value, because their sensitivity or specificity is inadequate.[22] Other causes of hypoglycemia should be properly investigated (see Differentials). For example, a morning cortisol level determination and/or adrenocorticotropic hormone (ACTH) stimulation testing should be performed if adrenal insufficiency is suspected. See a diagnostic algorithm for hypoglycemia below. Diagnostic algorithm. A systematic approach is often required to establish the true cause of hypoglycemia, using an algorithmic approach. Glucose and Insulin Levels During hypoglycemic episodes, patients should test their glucose at home to document hypoglycemia that is occurring with the episodes. Take into consideration that meter readings may not be accurate enough to establish the diagnosis. Test glucose and insulin levels simultaneously to document low glucose levels occurring in conjunction with inappropriate insulin levels. Keep in mind that whole blood glucose values may be spuriously low in polycythemia rubra vera because of the unequal distribution of glucose between erythrocytes and plasma, excessive glycolysis by erythrocytes, or both. Low blood glucose values in leukemia are due to excessive glycolysis by leukocytes and in hemolytic crisis from excessive glycolysis by nucleated erythrocytes. In the polycythemic patient or in serum of the leukemic or hemolytic patient, prompt measurement of glucose in plasma to which an antiglycolytic agent has been added should provide accurate results. A study by Craig and McLaughlin suggested that in patients with established post–bariatric surgery hypoglycemia, a blood glucose level below 54 mg/dL should indicate clinically important hypoglycemia. The investigators based this on the peak occurring in neuroglycopenic (NG) symptoms at glucose levels of < 54-40 mg/dL during provoked and real-world hypoglycemia, “the low sensitivity/high specificity of NG symptoms to detect hypoglycemia, and high prevalence of hypoglycemia unawareness at glucose values < 54 mg/dL.”[23] Oral glucose tolerance test Administer an oral glucose tolerance test if reactive hypoglycemia is suspected. An oral glucose tolerance test provides little benefit for the evaluation of fasting hypoglycemia. Perform the test for 5 hours while simultaneously testing glucose and insulin https://emedicine.medscape.com/article/122122-print 10/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print levels. To be meaningful, low blood sugar (< 50 mg/dL [< 2.78 mmol/L]) during the test should be accompanied by typical symptoms. Response to a mixed meal may be more representative. 72-Hour fasting plasma glucose A supervised fast is the most reliable diagnostic test for the evaluation of fasting hypoglycemia. Continue the fast for as long as 72 hours or until symptoms develop in the presence of hypoglycemia (blood sugar < 45 mg/dL (2.5 mmol/L) for women; < 55 mg/dL (3.05 mmol/L) for men). Obtain simultaneous insulin levels every 6 hours, when glucose is low and when symptoms develop. Also measure the beta-hydroxybutyrate serum level. Glucose and/or glucagon must be administered after blood sample withdrawal to abort hypoglycemic symptoms. The diagnosis of insulinoma is likely if the patient, at the conclusion of the fast, has neuroglycopenic symptoms, a fall in plasma glucose to less than 45 mg/dL (< 2.5 mmol/L), inappropriately elevated beta-cell polypeptides (insulin, proinsulin, and C-peptide levels), and a beta-hydroxybutyrate level of less than 2.7 mmol/L. For overnight fasting plasma glucose levels, symptoms of hypoglycemia may develop when the blood sugar is below 60 mg/dL (3.33 mmol/L). C-Peptide Levels Obtain C-peptide levels any time an elevated insulin level is obtained. Endogenous hyperinsulinemia from insulinoma is associated with elevated C-peptide concentrations with concurrent hypoglycemia. Exogenous hyperinsulinemia from injected insulin results in low concentrations of C-peptide, both because of the effect of the associated hypoglycemia and because of the direct suppressive effect of insulin on the pancreatic beta cell.[17] C-peptide levels are elevated in insulinoma, normal or low with exogenous insulin, and elevated with oral sulfonylureas. Radiologic Studies For the evaluation of insulinomas, computed tomography (CT) scanning and ultrasonography often are not helpful, because most of these tumors are small. Magnetic resonance imaging (MRI) may yield better results. Selective percutaneous transhepatic venous sampling often is helpful for localizing an insulinoma to the head, body, or tail of the pancreas, and selective arteriography is also often helpful in localizing insulin-secreting lesions. Octreotide scanning localizes insulinomas in approximately 50% of cases. Retroperitoneal tumors that are producing insulinlike growth factor (IGF) are usually imaged easily using a CT scan. Treatment Approach Considerations The mainstay of therapy for hypoglycemia is glucose. Other medications may be administered based on the underlying cause or the accompanying symptoms (not discussed here). Fasting hypoglycemia Dietary therapy may be effective for improving symptoms in patients with fasting hypoglycemia. Frequent meals/snacks are preferred, especially at night, with complex carbohydrates. If dietary therapy is inadequate, medical care for patients with fasting hypoglycemia may include intravenous (IV) glucose infusion. However, IV octreotide is effective for suppressing endogenous insulin secretion. Reactive hypoglycemia does not require medical care. Because exercise burns carbohydrates and increases sensitivity to insulin, patients with fasting hypoglycemia should avoid significant activity. However, patients with reactive hypoglycemia often find that their symptoms improve after embarking on a routine exercise program. https://emedicine.medscape.com/article/122122-print 11/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Definitive treatment for fasting hypoglycemia caused by a tumor is surgical resection. The success rate is good for benign isletcell adenomas, and the success rate for malignant islet-cell tumors can be as high as 50%. Reactive hypoglycemia For patients with reactive hypoglycemia, initiate a restriction of refined carbohydrates. Patients should avoid simple sugars, increase the frequency of their meals, and reduce the size of their meals. Patients may require 6 small meals and 2-3 snacks per day. Increased protein and fiber in the meal may be beneficial. In many patients, use of alpha-glucosidase inhibitors (acarbose and miglitol) may help. These medications cause reversible inhibition of pancreatic alpha-amylase and membranebound intestinal alpha-glucoside hydrolase enzymes. This enzyme inhibition results in delayed glucose absorption and a lowering of postprandial hyperglycemia and thus may prevent reactive hypoglycemia. Complications Hypoglycemia occurring as a complication of therapy for diabetes is common;[14, 15, 16, 17] in fact, mild hypoglycemia occurs in more than half of all patients with diabetes who are in therapy. Unrecognized infection causing hypoglycemia in patients with diabetes may result in recurrent hypoglycemic spells or progression of the infection. Acute sequelae of hypoglycemia can include coma, cardiac dysrhythmia, and death. The risk of permanent neurologic deficits increases with prolonged hypoglycemia; such deficits can include hemiparesis, memory impairment, diminished language skills, decreased abstract thinking capabilities, and ataxia. A study by Middleton et al suggested that sulfonylurea-related hypoglycemia can result in harmful cardiovascular effects. The study included 30 persons with sulfonylurea-treated type 2 diabetes, nine of whom were found to have hypoglycemia. The investigators found that five of the nine patients demonstrated hypoglycemia-associated QTc prolongation; additionally, the hypoglycemic individuals showed higher QT dynamicity relative to the other patients, even after the hypoglycemic events had passed.[24] Another study, by Mezquita-Raya et al, involving patients with type 1 or type 2 diabetes, also found a relationship between hypoglycemia and QTc prolongation, with the phenomenon occurring primarily in cases of type 1 diabetes. However, the results did not demonstrate a link between hypoglycemia and clinical arrhythmias in patients with type 1 or type 2 diabetes with high cardiovascular risk.[25] Untreated fasting hypoglycemia can lead to severe neuroglycopenia and, possibly, death. In an Australian study, Egi et al reported that in critically ill patients, hypoglycemia was found to be an independent risk factor for death, cardiovascular death, and infectious disease–related death.[26] In addition, there was a significant association between patient mortality rates and the severity of hypoglycemia.[26] A Japanese study, by Ohashi et al, indicated that in persons with type 1 or 2 diabetes being treated with insulin, nonsevere hypoglycemic events (NSHEs) can negatively affect diabetes management, daily activities, work, sleep, and the patient’s emotional state. Subjects in the study were aged 20 years or older.[27] Long-Term Monitoring Diabetic patients with episodes of hypoglycemia need education in nutrition, checking glucose levels at home, and early signs and symptoms of hypoglycemia. Recognition of early symptoms is paramount for self-treatment. Guidelines from a workgroup of the American Diabetes Association (ADA) and the Endocrine Society address the dangers of hypoglycemia in diabetic patients. The workgroup developed 2 tools for clinicians treating those with diabetes:a patient questionnaire and a provider checklist.[28, 29] The Hypoglycemia Patient Questionnaire includes the following questions: How well can you recognize the symptoms of low blood glucose? How often do you have hypoglycemic episodes? Have you needed assistance in the past during a hypoglycemic episode? Do you check your glucose level before driving? https://emedicine.medscape.com/article/122122-print 12/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Do those close to you know how to administer glucagon? The Hypoglycemia Provider Checklist is designed to verify that the clinician has asked the appropriate questions of the patient and has made the appropriate recommendations for managing low blood sugar.[28, 29] If the patient has fasting hypoglycemia and the cause is treatable, long-term follow-up usually is not needed. If the cause cannot be treated definitively (eg, inoperable pancreatic insulinoma), diazoxide can be used to elevate blood glucose levels and chemotherapy that specifically targets the beta cell (ie, using cytotoxic agents such as streptozotocin) should be considered. If the patient has reactive hypoglycemia, periodic outpatient monitoring is warranted to assess the continued presence of symptoms. Medication Medication Summary The mainstay of therapy for hypoglycemia is glucose. Other medications may be administered based on the underlying cause or the accompanying symptoms; however, these medications are not addressed in this article. Glucose Supplement Class Summary Glucose supplements are used to raise the patient's serum glucose. Dextrose (Glucose-D) Dextrose is a monosaccharide absorbed from intestine and distributed, stored, and used by tissues. Parenterally injected, dextrose is used in patients unable to obtain adequate oral (PO) intake. Direct oral absorption results in rapid increase of blood glucose concentrations. Dextrose is effective in small doses, and there is no evidence that it may cause toxicity. Concentrated dextrose infusions provide higher amounts of glucose and increased caloric intake with minimum fluid volume. The long-term management of hypoglycemia is dictated by its cause (eg, insulinoma). Glucose-Elevating Agents Class Summary Glucose-elevating agents can act in the pancreas or the peripheral tissues to increase blood glucose levels. Glucagon (GlucaGen, Gvoke) Pancreatic alpha cells of islets of Langerhans produce glucagon, a polypeptide hormone. This agent exerts effects opposite of insulin on blood glucose and elevates blood glucose levels by inhibiting glycogen synthesis and enhancing formation of glucose from noncarbohydrate sources, such as proteins and fats (gluconeogenesis). Glucagon also increases hydrolysis of glycogen to glucose (glycogenolysis) in the liver. This agent accelerates hepatic glycogenolysis and lipolysis in adipose tissue by stimulating cyclic AMP (cAMP) synthesis via adenylyl cyclase and enhancing https://emedicine.medscape.com/article/122122-print 13/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print phosphorylase kinase activity. Glucagon may be useful when intravenous (IV) access for dextrose administration is problematic. This agent may be administered as part of emergency medical services (EMS) protocol in patients with altered mental status and no IV access. It is also available as a ready-to-use subcutaneous (SC) solution in prefilled syringes or an autoinjector. Glucagon intranasal (Baqsimi) This agent activates hepatic glucagon receptors, which stimulate cyclic adenosine monophosphate (cAMP) synthesis. Hepatic glycogenolysis and gluconeogenesis are thus accelerated, with blood glucose levels consequently increasing. Glucagon requires preexisting hepatic glycogen stores to effectively treat hypoglycemia. Glucagon intranasal is indicated for severe hypoglycemic reactions in adults and children (aged 4 years or older) with diabetes. Inhibitors of insulin secretion Class Summary Agents that inhibit insulin secretion increase glucose levels by reducing peripheral glucose metabolism. Diazoxide (Proglycem) Diazoxide is a direct inhibitor of insulin secretion. This agent increases hepatic glucose output by inhibiting pancreatic insulin release and, possibly, through an extrapancreatic effect, as well as decreases cellular glucose uptake. Although, diazoxide has a very limited role in treating hypoglycemia, it can help improve symptoms of hypoglycemia caused by increased insulin secretion in patients awaiting surgery or those with nonresectable disease and may be indicated in some cases of insulinoma or overdosage with oral (PO) hypoglycemic agents. Hyperglycemic effect starts within 1 hour, lasting a maximum of 8 hours if the patient's renal function normal. Patients with refractory hypoglycemia may require high dosages. Octreotide (Sandostatin) This agent Inhibits insulin secretion. Octreotide acts primarily on somatostatin receptor subtypes II and V. It also inhibits growth hormone secretion and has a multitude of other endocrine and nonendocrine effects, including inhibition of glucagon, vasoactive intestinal peptides (VIP), and gastrointestinal peptides. Antineoplastic agents Class Summary Antineoplastic agents inhibit cell growth and proliferation. Streptozocin (Zanosar) Streptozocin has a high affinity for neuroendocrine cells, inhibits cell proliferation, and is cytolytic. This agent interferes with normal DNA function by alkylation and protein modification. Questions & Answers https://emedicine.medscape.com/article/122122-print 14/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Overview What is hypoglycemia (low blood sugar)? What is the focus of the medical history in the evaluation of hypoglycemia (low blood sugar)? How are the neurologic symptoms of hypoglycemia (low blood sugar) characterized? What signs and symptoms of reactive hypoglycemia? What is the significance of loss of consciousness in hypoglycemia (low blood sugar)? What are the signs and symptoms of gestational hypoglycemia? What is the Whipple triad in the diagnosis of hypoglycemia (low blood sugar)? What is included in the physical exam of suspected hypoglycemia (low blood sugar)? How does the presentation of hypoglycemia (low blood sugar) vary in elderly patients? What is the role of lab studies in the diagnosis of hypoglycemia (low blood sugar)? What is the role of imaging in the workup of hypoglycemia (low blood sugar)? Which procedures are performed in the workup of hypoglycemia (low blood sugar)? What is the mainstay of therapy for hypoglycemia (low blood sugar)? Which medications are used in the treatment of hypoglycemia (low blood sugar)? In addition to medications, what therapies are used in the management of hypoglycemia (low blood sugar)? When is surgery indicated in the treatment of hypoglycemia (low blood sugar)? What is hypoglycemia (low blood sugar)? How is hypoglycemia (low blood sugar) diagnosed? What is the pathophysiology of hypoglycemia (low blood sugar)? What causes hypoglycemia (low blood sugar)? What causes fasting hypoglycemia? Which drugs increase the risk of hypoglycemia (low blood sugar)? What is the role of opioid analgesics in the etiology of hypoglycemia (low blood sugar)? Which type 2 diabetes treatments increase the risk of hypoglycemia (low blood sugar)? What is factitious hypoglycemia? When should exogenous insulin be considered as the etiology of hypoglycemia (low blood sugar)? Which tumors cause hypoglycemia (low blood sugar)? Which insulin-producing tumors cause hypoglycemia (low blood sugar)? Which non-insulin producing tumors cause hypoglycemia (low blood sugar)? What are the causes of reactive hypoglycemia? What are congenital causes of hypoglycemia (low blood sugar)? Which conditions may cause hypoglycemia (low blood sugar)? What is the incidence of hypoglycemia (low blood sugar)? What is the incidence of insulin-producing tumors causing hypoglycemia (low blood sugar)? https://emedicine.medscape.com/article/122122-print 15/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print What are the age predilections for hypoglycemia (low blood sugar)? What is the prognosis of hypoglycemia (low blood sugar)? Presentation What should be the focus of medical history of a patient with hypoglycemia (low blood sugar)? What medical history suggests hypoglycemia (low blood sugar)? What should be the focus of social history of patients with hypoglycemia (low blood sugar)? What organ systems symptoms should be reviewed in the evaluation of hypoglycemia (low blood sugar)? What are neurologic symptoms of hypoglycemia (low blood sugar)? Why is the timing of onset of symptoms relative to meal ingestion significant in evaluation of hypoglycemia (low blood sugar)? What are characteristics of postprandial hyperglycemia in hypoglycemia (low blood sugar)? What is the association between hypoglycemia (low blood sugar) and dementia or cognitive impairment? What are risk factors for reactive hypoglycemia? What are risk factors for gestational hypoglycemia? Which dermatologic findings suggest hypoglycemia (low blood sugar)? Which physical findings are characteristic of hypoglycemia (low blood sugar)? Which head, eyes, ears, nose, and throat (HEENT) findings are characteristic of hypoglycemia (low blood sugar)? What are cardiovascular symptoms of hypoglycemia (low blood sugar)? What are respiratory symptoms of hypoglycemia (low blood sugar)? How are the symptoms presented in elderly patients with hypoglycemia (low blood sugar)? DDX Why is rapid diagnosis and treatment of hypoglycemia (low blood sugar) necessary? What may cause hypoglycemia (low blood sugar) in patients with diabetes? What are less common conditions that can cause hypoglycemia (low blood sugar)? Which conditions should be included in the differential diagnoses of hypoglycemia (low blood sugar)? What are the differential diagnoses for Hypoglycemia? Workup Which tests are performed in the workup of hypoglycemia (low blood sugar)? When is blood glucose tested in patients with hypoglycemia (low blood sugar)? What is the role of blood glucose testing in patients with hypoglycemia (low blood sugar)? What is the role of oral glucose tolerance testing in the diagnosis of hypoglycemia (low blood sugar)? What is the role of the 72-hour fasting plasma glucose test in the diagnosis of hypoglycemia (low blood sugar)? How are C-peptide levels used to diagnose hypoglycemia (low blood sugar)? What is the role of radiology in the diagnosis of hypoglycemia (low blood sugar)? Treatment What is the mainstay of therapy for hypoglycemia (low blood sugar)? https://emedicine.medscape.com/article/122122-print 16/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print What is the efficacy of dietary therapy in the treatment of fasting hypoglycemia? When is exercise indicated as treatment for fasting hypoglycemia? When is surgery indicated in the treatment of fasting hypoglycemia? What are treatment options for reactive hypoglycemia? How frequently is hypoglycemia (low blood sugar) a complication of diabetes? What are the acute sequelae of hypoglycemia (low blood sugar)? What complications can result from patients with sulfonylurea-related hypoglycemia? What are the complications of untreated fasting hypoglycemia? How does hypoglycemia (low blood sugar) affect the prognosis of critically ill patients? What is the effect of nonsevere hypoglycemic events (NSHEs) on diabetes management? How should episodes of hypoglycemia be managed in patients with diabetes? What guidelines have been published for the management of hypoglycemia (low blood sugar) in patients with diabetes? What is the Hypoglycemia Patient Questionnaire? What is the purpose of the Hypoglycemia Provider Checklist? When is long-term monitoring indicated for hypoglycemia (low blood sugar)? Medications Which medications are used for the treatment of hypoglycemia (low blood sugar)? Which medications in the drug class Glucose Supplement are used in the treatment of Hypoglycemia? Which medications in the drug class Glucose-Elevating Agents are used in the treatment of Hypoglycemia? Which medications in the drug class Inhibitors of insulin secretion are used in the treatment of Hypoglycemia? Which medications in the drug class Antineoplastic agents are used in the treatment of Hypoglycemia? Contributor Information and Disclosures Author Osama Hamdy, MD, PhD Medical Director, Obesity Clinical Program, Director of Inpatient Diabetes Program, Joslin Diabetes Center; Associate Professor of Medicine, Harvard Medical School Osama Hamdy, MD, PhD is a member of the following medical societies: American Association of Clinical Endocrinologists, American Diabetes Association Disclosure: Serve(d) as a director, officer, partner, employee, advisor, consultant or trustee for: on advisory panel of AstraZeneca Inc<br/>Received research grant from: USDA Dairy Council <br/>Have a 5% or greater equity interest in: HealthyMation Inc<br/>Received consulting fee from Merck Inc for teaching; Received consulting fee from Abbott Nutrition for consulting; for: Receieved consulting fee Sanofi Aventis for teaching. Coauthor(s) Vellore A R Srinivasan, MSc, PhD Professor of Biochemistry, Mahatma Gandhi Medical College and Research Institute, Sri Balaji Vidyapeeth University, India Disclosure: Received salary from Sri Balaji Vidyapeeth University, Mahatma Gandhi Medical College and Research Institute campus , Pondicherry ( Puducherry ) , India . P.C. 607 402 for employment. https://emedicine.medscape.com/article/122122-print 17/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print Kenneth J Snow, MD Associate Chief, Adult Diabetes, Joslin Clinic Kenneth J Snow, MD is a member of the following medical societies: American Association of Clinical Endocrinologists, American College of Endocrinology, American Diabetes Association, Endocrine Society Disclosure: Nothing to disclose. Chief Editor Romesh Khardori, MD, PhD, FACP Professor of Endocrinology, Director of Training Program, Division of Endocrinology, Diabetes and Metabolism, Strelitz Diabetes and Endocrine Disorders Institute, Department of Internal Medicine, Eastern Virginia Medical School Romesh Khardori, MD, PhD, FACP is a member of the following medical societies: American Association of Clinical Endocrinologists, American College of Physicians, American Diabetes Association, Endocrine Society Disclosure: Nothing to disclose. Acknowledgements Vasudevan A Raghavan, MBBS, MD, MRCP(UK) Director, Cardiometabolic and Lipid (CAMEL) Clinic Services, Division of Endocrinology, Scott and White Hospital, Texas A&M Health Science Center College of Medicine Vasudevan A Raghavan, MBBS, MD, MRCP(UK) is a member of the following medical societies: American College of Physicians-American Society of Internal Medicine, American Diabetes Association, American Heart Association, Endocrine Society, National Lipid Association, and Royal College of Physicians Disclosure: Nothing to disclose. David S Schade, MD Chief, Division of Endocrinology and Metabolism, Professor, Department of Internal Medicine, University of New Mexico School of Medicine and Health Sciences Center David S Schade, MD is a member of the following medical societies: American College of Physicians, American Diabetes Association, American Federation for Medical Research, Endocrine Society, New Mexico Medical Society, New York Academy of Sciences, and Society for Experimental Biology and Medicine Disclosure: Nothing to disclose. Francisco Talavera, PharmD, PhD Adjunct Assistant Professor, University of Nebraska Medical Center College of Pharmacy; Editor-in-Chief, Medscape Drug Reference Disclosure: Medscape Salary Employment References 1. Mathew P, Thoppil D. Hypoglycemia. 2018 Jan. [Medline]. [Full Text]. 2. Pugh SK, Doherty DA, Magann EF, et al. Does hypoglycemia following a glucose challenge test identify a high risk pregnancy?. Reprod Health. 2009 Jul 14. 6:10. [Medline]. [Full Text]. 3. Zhong VW, Juhaeri J, Cole SR, et al. Proximal HbA1C Level and First Hypoglycemia Hospitalization in Adults with Incident Type 2 Diabetes. J Clin Endocrinol Metab. 2019 Jan 3. [Medline]. 4. Riegger LQ, Leis AM, Golmirzaie KH, Malviya S. Risk Factors for Intraoperative Hypoglycemia in Children: A Multicenter Retrospective Cohort Study. Anesth Analg. 2020 Jun 25. [Medline]. 5. van Furth AM, de Heide LJM, Emous M, Veeger N, van Beek AP. Dumping Syndrome and Postbariatric Hypoglycemia: Supporting Evidence for a Common Etiology. Surg Obes Relat Dis. 2021 May 19. [Medline]. [Full Text]. 6. Hughes S. Pain Med Linked to Hypoglycemia. Medscape Medical News. Dec 11 2014. [Full Text]. 7. Fournier JP, Azoulay L, Yin H, et al. Tramadol Use and the Risk of Hospitalization for Hypoglycemia in Patients With Noncancer Pain. JAMA Intern Med. 2014 Dec 8. [Medline]. 8. Eriksson JW, Bodegard J, Nathanson D, Thuresson M, Nystrom T, Norhammar A. Sulphonylurea compared to DPP-4 inhibitors in combination with metformin carries increased risk of severe hypoglycemia, cardiovascular events, and all-cause mortality. Diabetes Res Clin Pract. 2016 Jul. 117:39-47. [Medline]. https://emedicine.medscape.com/article/122122-print 18/19 15/9/21 20:09 https://emedicine.medscape.com/article/122122-print 9. Gautier JF, Monguillon P, Verier-Mine O, et al. Which oral antidiabetic drug to combine with metformin to minimize the risk of hypoglycemia when initiating basal insulin?: A randomized controlled trial of a DPP4 inhibitor versus insulin secretagogues. Diabetes Res Clin Pract. 2016 Jun. 116:26-8. [Medline]. 10. Awad DH, Gokarakonda SB, Ilahi M. Factitious Hypoglycemia. StatPearls. 2021 Jan. [Medline]. [Full Text]. 11. Lamounier RN, Geloneze B, Leite SO, et al. Hypoglycemia incidence and awareness among insulin-treated patients with diabetes: the HAT study in Brazil. Diabetol Metab Syndr. 2018. 10:83. [Medline]. [Full Text]. 12. Hill NR, Thompson B, Bruce J, et al. Glycaemic risk assessment in children and young people with Type 1 diabetes mellitus. Diabet Med. 2009 Jul. 26(7):740-3. [Medline]. 13. Turnbull FM, Abraira C, Anderson RJ, et al. Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia. 2009 Aug 5. [Medline]. 14. Prolonged Nocturnal Hypoglycemia Is Common During 12 Months Of Continuous Glucose Monitoring In Children And Adults With Type 1 Diabetes. Diabetes Care. 2010 Mar 3. [Medline]. [Full Text]. 15. Swinnen SG, Dain MP, Aronson R, et al. A 24-week, randomized, treat-to-target trial comparing initiation of insulin glargine oncedaily with insulin detemir twice-daily in patients with type 2 diabetes inadequately controlled on oral glucose-lowering drugs. Diabetes Care. 2010 Mar 3. [Medline]. 16. Ito T, Otsuki M, Igarashi H, et al. Epidemiological Study of Pancreatic Diabetes in Japan in 2005: A Nationwide Study. Pancreas. 2010 Feb 22. [Medline]. 17. Chen L. A literature review of intensive insulin therapy and mortality in critically ill patients. Clin Nurse Spec. 2010 Mar-Apr. 24(2):806. [Medline]. 18. Garza H. Minimizing the risk of hypoglycemia in older adults: a focus on long-term care. Consult Pharm. 2009 Jun. 24 Suppl B:1824. [Medline]. 19. Boucai L, Southern WN, Zonszein J. Hypoglycemia-associated Mortality Is Not Drug-associated but Linked to Comorbidities. Am J Med. 2011 Nov. 124(11):1028-35. [Medline]. [Full Text]. 20. Feil DG, Rajan M, Soroka O, et al. Risk of hypoglycemia in older veterans with dementia and cognitive impairment: implications for practice and policy. J Am Geriatr Soc. 2011 Dec. 59(12):2263-72. [Medline]. 21. Parikh NS, Ahlawat R. Glycogen Storage Disease Type I (Von Gierke Disease). 2018 Jan. [Medline]. [Full Text]. 22. Lin YY, Hsu CW, Sheu WH, Chu SJ, Wu CP, Tsai SH. Use of therapeutic responses to glucose replacement to predict glucose patterns in diabetic patients presenting with severe hypoglycaemia. Int J Clin Pract. 2009 Aug. 63(8):1161-6. [Medline]. 23. Craig CM, McLaughlin TL. Defining clinically important hypoglycemia in patients with postbariatric hypoglycemia. Surg Obes Relat Dis. 2021 Jun 29. [Medline]. 24. Middleton TL, Wong J, Molyneaux L, et al. Cardiac Effects of Sulfonylurea-Related Hypoglycemia. Diabetes Care. 2017 Feb 21. [Medline]. 25. Mezquita-Raya P, Reyes-Garcia R, de Torres-Sanchez A, Matarin MG, Cepero-Garcia D, Perez de Isla L. Electrical changes during hypoglycaemia in patients with type 1 and type 2 diabetes and high cardiovascular risk. Diabetes Res Clin Pract. 2018 Jan 31. 138:44-6. [Medline]. 26. Egi M, Bellomo R, Stachowski E, et al. Hypoglycemia and outcome in critically ill patients. Mayo Clin Proc. 2010 Mar. 85(3):217-24. [Medline]. [Full Text]. 27. Ohashi Y, Wolden ML, Hyllested-Winge J, Brod M. The diabetes management and daily functioning burden of non-severe hypoglycaemia in Japanese people treated with insulin. J Diabetes Investig. 2017 Feb 8. [Medline]. [Full Text]. 28. New Guidelines Stress Hypoglycemia Affects All Diabetics. Medscape Medical News. Available at http://www.medscape.com/viewarticle/802949?nlid=30763_1621. Accessed: May 2, 2013. 29. Seaquist ER, Anderson J, Childs B, Cryer P, Dagogo-Jack S, Fish L, et al. Hypoglycemia and diabetes: a report of a workgroup of the american diabetes association and the endocrine society. Diabetes Care. 2013 May. 36(5):1384-95. [Medline]. [Full Text]. 30. Kong AP, Chan JC. Hypoglycemia and Comorbidities in Type 2 Diabetes. Curr Diab Rep. 2015 Oct. 15 (10):646. [Medline]. https://emedicine.medscape.com/article/122122-print 19/19