Effect of the Sequence of the Thermoelectric Generator and the Three-Way Catalytic Converter on Exhaust Gas Conversion Efficiency. NANDO pdf
Anuncio

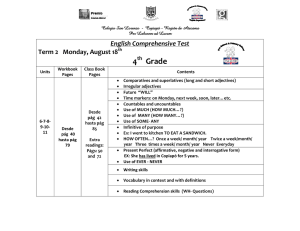
Journal of ELECTRONIC MATERIALS, Vol. 42, No. 7, 2013 DOI: 10.1007/s11664-012-2454-2 Ó 2013 TMS Effect of the Sequence of the Thermoelectric Generator and the Three-Way Catalytic Converter on Exhaust Gas Conversion Efficiency CHUQI SU,1 NAIQIANG TONG,1,2 YUMAN XU,1 SHAN CHEN,1 and XUN LIU1 1.—Hubei Key Laboratory of Advanced Technology for Automotive Components, Automobile Engineering Institute, Wuhan University of Technology, Hongshan District, 205 Luoshi Road, Wuhan 430070, China. 2.—e-mail: [email protected] The potential for thermoelectric exhaust heat recovery in vehicles has increased with recent improvements in the efficiency of thermoelectric generators (TEGs). The problem with using thermoelectric generators for vehicle applications is whether the device is compatible with the original vehicle exhaust system, which determines the quality of the exhaust gas treatment and the realization of energy conservation and emission reduction. Based on ANSYS CFX simulation analysis of the impact of two positional relationships between the TEG and three-way catalytic converter in the exhaust system on the working efficiency of both elements, it is concluded that the layout with the front three-way catalytic converter has an advantage over the other layout mode under current conditions. New ideas for an improvement program are proposed to provide the basis for further research. Key words: Thermoelectric generator (TEG), three-way catalytic converter, impact on efficiency, ANSYS CFX simulation analysis INTRODUCTION Currently, about 30% of the total energy from automotive engines is used to drive the vehicle, whereas another 30% is consumed by the cooling system, and the remaining 40% is emitted as heat by the exhaust system. A normal sedan wastes 20 kW to 30 kW of heat at uniform speed when the engine is under common working conditions.1 Part of this waste heat can be recovered and reused to reduce the fuel consumption of the vehicle as well as environmental pollution. Automobile exhaust waste heat thermoelectric power generation systems can be used on vehicles to recover part of this energy. Although not reaching zero emissions, such systems are appropriate for the current state of energy recuperation technology.2,3 An automobile exhaust waste heat thermoelectric power generation system consists of a hot-side heat (Received July 7, 2012; accepted December 28, 2012; published online February 9, 2013) exchanger, a cold-side water tank, and a clamping device.4 Thermoelectric modules made of semiconductor material are placed between the hot-side heat exchanger that is connected to the engine exhaust pipe and the cold-side water tank, enabling the conversion of the temperature difference between the hot and cold side into power.5 Based on the Seebeck effect, thermoelectric modules can make use of a difference in temperature between their two ends to generate power. In this work, we use low-temperature modules made of Ti2Be3, which are capable of withstanding hot temperatures below 300°C.6 However, such addition of a thermoelectric generator (TEG) to the automotive exhaust system will affect the overall system, especially the three-way catalytic converter.7 Since the TEG and three-way catalytic converter perform properly when the temperature reaches a certain level, simulation analysis of the relative positional relationship of these two elements was carried out to determine a reasonable layout. This research also provides the 1877 1878 Su, Tong, Xu, Chen, and Liu foundation for future improvement programs. study directions and ANALYSIS ON THE RELATIVE POSITIONAL RELATIONSHIP BETWEEN THE TEG AND THREE-WAY CATALYTIC CONVERTER There are two positional relationship types: Fig. 1 shows one type in which the TEG is placed in front of the three-way catalytic converter, in which the exhaust gas goes through the TEG first and then the three-way catalytic converter; Fig. 2 shows the other type in which the TEG is placed behind the three-way catalytic converter, in which exhaust gas goes through the three-way catalytic converter first and then the TEG. When the TEG is in front, it absorbs some heat from the exhaust gas. Consequently, the remaining heat of this gas is insufficient to raise the temperature of the three-way catalytic converter to the required standard; when the three-way catalytic converter is in front, the temperature of the exhaust gas rises after the reaction in the three-way catalytic converter. As a result, the temperature of the hot side of the TEG exceeds the maximum upper temperature of the thermoelectric modules, possibly damaging them. According to the characteristics of the two layout modes discussed above, the following assumptions are made: 1. When the TEG is in front, one analyzes whether the temperature of the exhaust gas could reach the standard value demanded by the three-way catalytic converter, regardless of heat release in the catalytic reaction; 2. When the three-way catalytic converter is in front, one analyzes whether the TEG can work normally using the hotter gas after its passage through the three-way catalytic converter. SIMULATION Simulation of the TEG in Front Layout Boundary Condition Setting The popular computational fluid dynamics (CFD) software ANSYS CFX was adopted for the simulation. The boundary conditions must be specified, and in this case are set as follows: Fig. 1. Model with the TEG first. Inlet boundary condition: average velocity of exhaust, 15 m/s; exhaust temperature, 800 K. Outlet boundary condition: static pressure, 0 MPa. To simplify the simulation model, it assumed in this case that the outlet is connected to the surrounding environment and the air pressure is equal to atmospheric pressure correspondingly. Convective heat transfer coefficient of the surface of the heat exchanger of the TEG: 20 W/(m2 K). The three-way catalyst parameters under the assumption that the reaction therein is uniform are: the medium is set as common ceramic materials; energy source value, 1 9 106 W/m3; convective heat transfer coefficient, 10 W/(m2 K). Gas in this system is considered incompressible, the flow is fully turbulent, and the molecular viscosity can be assumed negligible in the simulation model. All of these are in line with the application of k–e turbulence model equations. Simulation Results Figures 3–5 show the results obtained from the CFX simulations. Figure 3 presents the temperature distribution of the layout with the TEG in front. The highest temperature on the surface of the heat exchanger is 540.7 K (282.6°C), and the lowest one is 458.6 K (237°C). Due to the exhaust speed, which is set as 15 m/s, and the exhaust direction from the heat exchanger to the three-way catalyst, the effect caused by the downstream three-way catalyst on the convective heat transfer of the upstream heat exchanger is slight. The surface heat flux density distribution of the whole model is shown in Fig. 4. Negative values indicate that the surface of the model is exothermic, and the value represents the heat flux density. The distribution of the heat flux density is similar to that of the temperature distribution. The higher the temperature, the larger the heat flux density value. The average heat flux density of the available top and bottom surface of the heat exchanger based on the calculation results is 3910 W/m2. Figure 5 shows the temperature of the medium of the three-way catalyst. Because of the in-front position of the heat exchanger, the exhaust temperature of the three-way catalyst inlet is low, as is Fig. 2. Model with the three-way catalyst first. Effect of the Sequence of the Thermoelectric Generator and the Three-Way Catalytic Converter on Exhaust Gas Conversion Efficiency 1879 Fig. 3. Temperature distribution with the TEG first. Fig. 4. Heat flux distribution with the TEG first. Fig. 5. Three-way catalyst carrier temperature distribution with the TEG first. the whole temperature of the three-way catalyst. The highest temperature is 260°C, which occurs at the center of the medium inlet; correspondingly the lowest temperature is 164°C, locating at the end of the medium and near the three-way catalyst shell. The average temperature of the whole medium is 230°C, which is lower than the ignition temperature (250°C) of the harmful gas in the vehicle exhaust. This means that, when the vehicle exhaust flows through the medium of the three-way catalyst, the catalytic reaction only happens in some of the medium rather than uniformly throughout the 1880 Su, Tong, Xu, Chen, and Liu Fig. 6. Temperature distribution with the three-way catalyst first. Fig. 7. Heat exchanger local temperature distribution with the three-way catalyst first. whole medium. It is easy to conclude that the layout with the TEG in front is unfavorable for normal operation of the three-way catalyst. Simulation of the Three-Way Catalyst in Front Layout Simulation Results Figure 6 presents the temperature distribution of the system with the three-way catalyst in front as obtained from the CFX simulations. Due to the in-front location of the three-way catalyst and the large amount of heat released by the catalytic reaction therein, high-temperature areas are centralized at the shell of the three-way catalyst. Meanwhile, the heat exchanger is located downstream in the exhaust system and does not have a heat source. The temperature of the heat exchanger is low and changes little; the temperature distribution is shown in Fig. 7. The highest temperature on the surface of the heat exchanger is 510 K (265.6°C), and the lowest one is 439 K (226.1°C). The highest temperature is not higher than the maximum temperature that the thermoelectric modules can withstand, and is even Fig. 8. Model of the TEG with two heat exchangers. lower than for the layout with the TEG in front. The average heat flux density of the available top and bottom surface of the heat exchanger based on the calculation results is 3510 W/m2. Thus, the heat emission of the chemical reaction in the three-way catalyst is less than the heat dispersed by the exhaust. The largest useful surface area of one surface of the heat exchanger is about 0.15 m2. According to the simulation analysis, when the TEG is in front, the total energy exchange of the heat exchanger surfaces is 1173 W; the temperature of the threeway catalyst is only 230°C under this condition, which cannot meet the normal working requirements for the three-way catalyst. Effect of the Sequence of the Thermoelectric Generator and the Three-Way Catalytic Converter on Exhaust Gas Conversion Efficiency 1881 Fig. 9. Heat flux distribution of the TEG with two heat exchangers. When the three-way catalyst is in front, it can perform properly, but the total energy of the TEG heat exchanger’s surfaces is 1053 W, which is lower than the previous condition by 120 W. Optimization of the Three-Way Catalyst in Front Layout In the three-way catalyst in front system, the heat emitted by the chemical reaction in the three-way catalyst is less than the heat dispersed by the exhaust. Thus, less heat can be recovered by the TEG located in the downstream exhaust system. Expanding the area for heat transfer is an effective way to strengthen the heat convection, so it is practicable to increase the number of heat exchangers in the TEG in order to recover more exhaust heat. In this study, a three-dimensional model of a TEG with two heat exchangers connected by a Y-type fitting was designed as shown in Fig. 8. Considering the restricted computational capacity available, only a double-layer heat exchanger was simulated. The temperature and velocity of the outlet gas of the three-way catalyst are regarded as the inlet boundary conditions of the double-layer heat exchanger, with the other boundary conditions kept unchanged. The simulation result is shown in Fig. 9. The simulation results indicate that the heat flux density of each available surface of the heat exchanger is basically consistent, and the average surface heat flux is 2200 W/m2. Although the average heat flux density of a single surface of the double-layer heat exchanger is less than that of the single-layer heat exchanger, the total energy recovery is increased by 25%. In comparison with the heat recovered by the system with the singlelayer heat exchanger in front, the heat converted by the system with the three-way catalyst with doublelayer heat exchanger in front is increased by 12.5%. CONCLUSIONS The challenge with onboard TEG use is to ensure that the three-way catalyst and TEG can operate normally at their corresponding working temperatures. According to the presented simulation analysis, the results for the two considered layouts are as follows: 1. With the TEG in front, the three-way catalyst cannot work normally, as its temperature is lower than its working temperature, while the TEG can recover more energy because of the high temperature. 2. If the three-way catalyst is in front, it will work normally. Although the chemical reactions give off a large amount of heat, the temperature at the TEG is lower than the condition with the TEG in front. The heat recoverable through the thermoelectric power generation system is lower than in the first condition. Therefore, each of the two design has its own advantages. In the existing conditions, the layout with the TEG after the three-way catalyst seems more practical. For both conditions, changing the materials of the catalytic carrier and the heat exchanger or increasing the number of heat exchangers will improve the thermal energy recovery efficiency. These measures will be studied in future research. ACKNOWLEDGEMENTS This work is funded by Grant No. 2007CB607501 from International Science &Technology Cooperation Program of China, No. 2011DFB60150 developed by General Motors Corporation and Wuhan University of Technology. REFERENCES 1. F.Stabler, DARPA/ONR/DOE High Efficiency Thermoelectric Workshop (2002), p. 1. 2. K.T. Chau and Y.S. Wong, Energy Convers. Manage. 43, 1953 (2002). 3. K.T. Chau and Y.S. Wong, Energy Convers. Manage. 42, 1059 (2001). 4. J. Vazquez, et al., Proceedings of 7th European Workshop on Thermoelectrics (Pamplona, Spain, 2002). 5. D.M. Rowe and G. Min, J. Power Sources 73, 193 (1998). 6. X. Niu, et al., J. Power Sources 188, 621 (2009). 7. S.E. Votzs, C.R. Morgan, D. Liederman, and S.M. Jacob, Ind. Eng. Chem. Res. 12, 194 (1973). Copyright of Journal of Electronic Materials is the property of Springer Science & Business Media B.V. and its content may not be copied or emailed to multiple sites or posted to a listserv without the copyright holder's express written permission. However, users may print, download, or email articles for individual use.