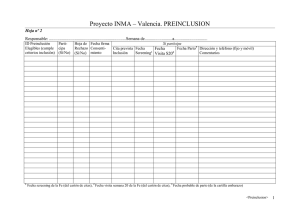
10-02-25 GuiaScreening v3 English
Anuncio

Preclinical Research in Medicines Directory of Screening Platforms and Chemical Libraries in Spain March 2010 La Plataforma Tecnológica Española Medicamentos Innovadores cuenta con apoyo financiero del Ministerio de Ciencia e Innovación y de los Fondos FEDER Directory of Screening Platforms and Chemical Libraries in Spain Index INTRODUCTION SUMMARY PLATFORMS LIBRARIES La Plataforma Tecnológica Española Medicamentos Innovadores cuenta con apoyo financiero del Ministerio de Ciencia e Innovación y de los Fondos FEDER Ref. Expdte: RET-090000-2008-001 Cuantía financiada: 235.500€ (anualidades 2008-2009) -2- Directory of Screening Platforms and Chemical Libraries in Spain INTRODUCTION This document is the first edition of the Directory of Screening Platforms and Chemical Libraries localized in Spain. The current version has been developed with the collaboration of 19 Screening Platforms and 4 Chemical Libraries within the planned activities of the Spanish Technological Platform for Innovative Medicines (PTEMI) and with the collaboration of the Spanish Association for the Pharmaceutical Industry in Spain, FARMAINDUSTRIA. The main objective of the Spanish Technological Platform for Innovative Medicines is to promote the collaborative research between the different stakeholders in the pharmaceutical sector in Spain. Between the specific objectives of this Directory are to stimulate the realization of preclinical research in medicines in Spain as well as to make an attractive and useful tool available for the pharmaceutical companies during the drug development project. This Directory has been published in early 2010 and will have a complete diffusion among all kinds of organizations interested in Preclinical Research. A questionnaire was distributed to 19 Screening Platforms and 4 Chemical Libraries in Spain during January and December 2009 to collect data for this Directory. The consulted organizations have validated all the information collected in this Directory. The Directory is divided in two main sections, the first one offering a general view of the Screening Platforms and Chemical Libraries gathered in the Directory, and the second presenting each Screening Platform or Chemical Library with complete descriptions and individual facilities organized as shown below. Screening Platforms Chemical Libraries General Information General Information Installations Installations Management of chemical libraries Management of chemical libraries for assays Assays available Management of own chemical libraries Production of biological reagents Biological/pharmacological annotation of the chemical library Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered -3- Directory of Screening Platforms and Chemical Libraries in Spain SUMMARY The table below depicts the Screening Platforms and Chemical Libraries included in this Directory: Screening Platforms (Full name) Short name Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) MEDINA BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna BIOLAB APOINTECH, S.L. APOINTECH Screening Biomar; Instituto Biomar, S.A. PSBIOMAR Biomed Division – Leitat Tehnological Center BIOMED TM BIOPLATFORMSCREEN (Thrombotargets S.L.) BIOPLAT NEUROSCIENCE TECHNOLOGIES S.L.P. NEUROSC Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) PSFUSEF NEUROLAB_IQM PSNEUROLAB MEJORAN LAB AUTOMATION S.L. MEJORAN Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN RMNCNIO Vivia Biotech SL VIVIA BIOBIDE, S.L. BIOBIDE FUNDACIÓN GAIKER GAIKER INNOPROT S.L. INNOPROT Pharmakine S.L. PHARMAKINE Biopolis S.L. BIOPOLIS Plataforma de Screening in vivo en Drosophila melanogaster, VALENTIA BioPharma VALENTIA Fundación Centro de Investigación Príncipe Felipe CIPF -4- Directory of Screening Platforms and Chemical Libraries in Spain Chemical Libraries (Full name) Short name Quimiotecas Biomar; Instituto Biomar, S.A. QBIOMAR Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) CHEMBIO NEUROLAB_IQM (Instituto de Química Médica CSIC) QNEUROLAB Fundación Centro de Investigación Príncipe Felipe CIPF-Q -5- Directory of Screening Platforms and Chemical Libraries in Spain Location of the Screening Platforms (PS) and Chemical Libraries (Q) PS BIOBIDE PS GAIKER PS INNOPROT PS PSFUSEF País Vasco PS PHARMAKINE PS BIOMED PS BIOPLAT PS APOINTECH PS NEUROSC PS BIOMAR Q CHEMBIO Q BIOMAR PS BIOMED PS NEUROLAB I. Baleares PS MEJORAN PS BIOPOLIS PS VALENTIA PS CIPF Q CIPF-Q PS RMNCNIO PS VIVIA Q NEUROLAB PS MEDINA PS BIOLAB -6- Directory of Screening Platforms and Chemical Libraries in Spain Summary Main conclusions derived from the present directory are: • Out of the 19 Screening Platforms included, 12 are privately owned and the remaining 7 are associated to the national public research system. Out of the 4 Chemical Libraries included, 3 are associated to the national public research system and the remaining is privately owned. • The first Screening Platform was found in 1996 (the Foundation GAIKER), and further Screening Platforms have been established through till 2009 (the Fundación MEDINA). In general, most of the Screening Platforms have been established during the last five years. • The total usable space of the Screening Platforms amounts to 11,094 square meters (m2), with an average of 584 m2 each, although the size can range from 15 m2 to 3,250 m2. The total usable space of the Chemical Libraries amounts to 2,505 m2. • Out of the 19 Screening Platforms included, 14 of them are able to manage chemical libraries for assays having a capacity to store compounds ranging from 500.000 (Fundación MEDINA and Vivia Biotech SL) to 60 compounds (Fundación GAIKER) • However, only 9 Screening Platforms have own Chemical Library, the size of these can range from 100.000 compounds (Fundación MEDINA) to 98 compounds (APOINTECH, S.L.) • All the Screening Platforms describe the assays available inside the organization and 15 of them have capacity to produce biological reagents. • There is a total of 278 Staff or personnel working in the 19 Screening Platforms, 208 of which are permanent employees and 70 have specific-term contracts. Each platform would have an average of 14,6 employees, of them 10,9 are permanent and 3,7 contracted employees. • Out of the 19 Screening Platforms included, only 4 of them have got any accreditation by an official Agency or have passed any audit. Another six platforms are in the process to get an accreditation. • All the Screening Platforms except one have screening experience and all of them describe in detail the available capacity offered for services or for collaboration. • The storage capacity of compounds in the Chemical Libraries included in the Directory ranges from 50,000 to 2,000 compounds. Two of the Chemical Libraries have annotated biological or pharmacological their library. -7- Directory of Screening Platforms and Chemical Libraries in Spain Ownership and when established Screening Platforms Ownership Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Public 2009 BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Public 2004 APOINTECH, S.L. Private 2004 Screening Biomar; Instituto Biomar, S.A. Private 1996 Biomed Division – Leitat Tehnological Center Private 2008 Private 2006 NEUROSCIENCE TECHNOLOGIES S.L.P. Private 2005 Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Public 2004 NEUROLAB_IQM Public 2004 MEJORAN LAB AUTOMATION S.L. Private 2008 Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Public 2008 Vivia Biotech SL Private 2007 BIOBIDE, S.L. Private 2005 FUNDACIÓN GAIKER Public 1996 INNOPROT S.L. Private 2007 Pharmakine S.L. Private 2002 Biopolis S.L. Private 2003 Plataforma de Screening in vivo en Drosophila melanogaster, VALENTIA BioPharma Private 2006 Fundación Centro de Investigación Príncipe Felipe Public 2005 TM BIOPLATFORMSCREEN (Thrombotargets S.L.) -8- Established Directory of Screening Platforms and Chemical Libraries in Spain Ownership and when established Chemical Libraries Ownership Quimiotecas Biomar; Instituto Biomar, S.A. Private 1996 Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) Public 2007 NEUROLAB_IQM (Instituto de Química Médica CSIC) Public 1997 Fundación Centro de Investigación Príncipe Felipe Public 2005 -9- Established Directory of Screening Platforms and Chemical Libraries in Spain Usable space and management of chemical libraries Usable surface (m2) Chemical library for assays Capacidad (nº compounds) Own chemical library Number of compounds MEDINA 3.250 YES 500,000 SÍ 100,000 BIOLAB 50 YES 15,000 SÍ 1,532 APOINTECH 174 YES 10,000 YES 98 PSBIOMAR 2.300 - - YES 42,000 BIOMED 200 YES 1,000 NO - BIOPLAT 80 YES 50,000 NO - NEUROSC 120 YES 10,000 NO - PSFUSEF 250 YES 20,000 YES 12,000 PSNEUROLAB 15 YES 100 YES 1,000 MEJORAN 200 NO - NO RMNCNIO 30 YES 2,000 YES 380 VIVIA 400 YES 500,000 YES 2,840 BIOBIDE 1.000 YES 1,000 NO - GAIKER 450 YES 60 NO - INNOPROT 300 NO - NO - PHARMAKINE 525 NO - NO - BIOPOLIS 1.500 NO - NO - VALENTIA 100 YES 100,000 NO - CIPF 150 YES 14,000 YES 14,056 Platforms - 10 - Directory of Screening Platforms and Chemical Libraries in Spain Usable space and management of chemical libraries Usable surface (m2) Number of compounds Biological or pharmacological annotated QBIOMAR 2,300 42,000 YES CHEMBIO 50 50,000 NO QNEUROLAB 5 2,000 NO 150 14,056 YES Chemical libraries CIPF-Q - 11 - Directory of Screening Platforms and Chemical Libraries in Spain When established and usable space 4 Screening Platforms Established 3.250 3 2 3 2 Usable space of Screening Platforms (m2) 2.300 2 1.500 1 1 1 1.000 250 300 150 174 200 200 120 100 80 15 30 50 1996 1997 1998 1999 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 - 12 - 525 400 450 Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents, accreditations, personnel and screening experience Production of biological reagents Accreditations and audits Personnel Permanent Contracted Screening experience MEDINA YES In process 23 18 5 YES BIOLAB YES NO 6 6 YES APOINTECH YES YES 11 2 9 YES PSBIOMAR YES - 40 30 10 YES BIOMED YES YES 11 11 YES BIOPLAT NO NO 3 3 YES NEUROSC YES In process 11 8 3 YES PSFUSEF YES In process 13 2 11 YES PSNEUROLAB NO NO 2 2 YES MEJORAN NO NO 10 10 NO RMNCNIO YES NO 2 2 YES VIVIA NO NO 24 22 2 YES BIOBIDE YES YES 19 17 2 YES GAIKER YES YES 29 17 12 YES INNOPROT YES In process 11 11 YES PHARMAKINE YES Internally 16 16 YES BIOPOLIS YES In process 33 33 YES VALENTIA YES In process 11 6 CIPF YES NO 3 Platforms - 13 - 5 YES 3 YES Directory of Screening Platforms and Chemical Libraries in Spain SCREENING PLATFORMS Andalucía Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Canarias BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Castilla y León APOINTECH, S.L. Castilla y León Screening Biomar; Instituto Biomar, S.A. Cataluña Biomed Division – Leitat Tehnological Center Cataluña BIOPLATFORMSCREENTM (Thrombotargets S.L.) Cataluña NEUROSCIENCE TECHNOLOGIES S.L.P. Galicia Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Madrid NEUROLAB_IQM Madrid MEJORAN LAB AUTOMATION S.L. Madrid Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Madrid Vivia Biotech SL País Vasco BIOBIDE, S.L. País Vasco FUNDACIÓN GAIKER País Vasco INNOPROT S.L. País Vasco Pharmakine S.L. - 14 - Directory of Screening Platforms and Chemical Libraries in Spain SCREENING PLATFORMS (continue) C. Valenciana Biopolis S.L. C. Valenciana Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma C. Valenciana Fundación Centro de Investigación Príncipe Felipe CHEMICAL LIBRARIES Castilla y León Quimiotecas Biomar; Instituto Biomar, S.A. Cataluña Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) Madrid NEUROLAB_IQM (Instituto de Química Médica CSIC) C. Valenciana Fundación Centro de Investigación Príncipe Felipe Quimioteca - 15 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 16 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) General Information Name and ownership Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA). Ownership public–private. Start-up date MEDINA was created on November 18, 2008, and stemmed from the former Merck Basic Research Center in Madrid, Spain (Merck-CIBE 1952-2008). As of the end of 2009, the assets have been transferred and Medina is currently a fully operating center. Contact information Name Olga Genilloud, PhD. Scientific Director Postal address Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Parque Tecnológico de las Ciencias de la Salud Avda de Conocimiento 3 18100 GRANADA, España E-mail [email protected] Phone number +34 958 993965 - 17 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Installations Usable surface area 3.250 m2 Authorization from Nuclear Safety Authority Use of the Radiactive facilities of the University of Granada (if necessary) Location: Edificio Centro Desarrollo Farmacéutico y Alimentario, Parque Tecnológico de las Ciencias de la Salud Other (specify) - 18 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Management of chemical libraries Management of chemical libraries for assays Storage capacity for compounds (number) 500.000 in 96 well plates Storage conditions (temperature, concentration of solvents,...) -20ºC and room temperature 100% DMSO; DMSO/AQ 20%/80%; 100%AQ Minimum volume accepted 1-50 ųl, 5-500 ųl, 500-1000 ųl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library Tubes + 96 well plates, Others: Reformattable Coding method used in the chemical library Bar code, alphanumerical, Oracle data base State of compounds in the chemical library Solution: 1xWholeBrothEquivalent/ 10mg/mL/ DMSO 20% Number of compounds 100.000 crude biological extracts from microbial natural products Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library 500 ± 200 D (Estimation: Current Chemical Biology, 2007, 1, 115-127) Library of crude biological extracts Average number of H donors and acceptors 30% (Current Chemical Biology, 2007, 1, 115-127) Percentage of natural products 100,00% Does the library contain mixtures of natural products? YES Storage conditions (temperature, atmosphere, light) -20º, in absence of light Degree of purity of compounds in the chemical library (mean value) Complex mixtures Exclusion criterion for purity (% cut-off) N/A - 19 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Targets available GPCRs Ion channels Peptide receptors Receptors with enzymatic activity Kinases Phosphatases Transporters Other Targets: antibacterial, antifungal, parasitic diseases (malarie) Types of assay Radioactive: Filtrate SPA Flashplate Luminiscence Colorimetric Fluorescence TRF FRET No-Automated patch-clamp HPLC/MS/MS HCS HTS in bacterial, fungal and parasite (malaria) Others: Agar diffusion Assay format 48, 80, 96 and 384 well plates - 20 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Biotransformations Growth conditions Monolayer Spinner flasks Roller Bottles Other: Robotic cell culturing, SelecT Production capacity for biological reagents (nº of experimental points/week) SelecT: 30.000-40.000 data points / week - 21 - Directory of Screening Platforms and Chemical Libraries in Spain Infrastructure Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Description (robotized systems, detectors, etc) • 1x SelecT • 1x Typhoon • 5x Biomek FX • 2x Imagen Analyzer • 3x EP3 • 2x Patch Clamp • 4x Aquarius • 1x API4000 • 6x Tecan • 1x Micromass Quatro • 2x Tecan Ultraevolution • 1x Bioteck Washer • 3x MultiProbes • 1x Genevac HT-24 • 2x Victor II • 1x Genevac HT-8 • 2x Espectrafluor • • 2xVIPR • 1x Gilson HPLC • 2x CEDES • 2x ISCO combiflash • 1x Matrix Cell mate • 1x Vellocity 11 Benchcell-Labeler-Sealer • Thermo Multidrop (varias unidades) • 2x Agilent 1100+MSD (HPLC/MS) • 1x Flexdrop • • 2x TopCount - 22 - 1x Bruker Maxis (High-res uPLC/MS) 21x Khüners Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Personnel Fixed contract Nº % Doctors 5 100 Postgraduates 7 Qualified (diploma level) Function Temporary contract Nº % Function Scientific direction and R+D 1 50 R+D 100 R+D 4 100 R+D 3 100 R+D Technicians 1 100 Technician Other personnel (specify) 2 100 Business Manager and administration - 23 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Validation criteria per plate assayed Edge effect, Controls, QC, CV, SD, Z factor, Z score Validation criteria per screening survey Z’ Factor, SD, Edge effect Validation criteria concentration-response curves Controls, IC50, DE50, Vindow, inflection point Software for analysis of results JMP7, Sofware developed internally, Genedata, Nautilus, XLfit Database for storage and consultation of results Oracle Accreditations and audits available GLPs, ENAC and ISO9001 the implementation process is in progress Center endorsed by its previous experience at Farmaindustria - 24 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) Screening experience Experiencia en Merck - CIBE Year 2004 2005 2006 2007 2008 (2009) 5 6 8 7 8 # Contracts with industry (and size) N/A N/A N/A N/A N/A # Screening campaigns 14 14 16 18 20 # 2000 2200 2500 2750 3000* # 12 14 16 18 20 # Research projects Maximum yield per day Assays developed/optimized * Referred Data from the evaluation of ADME/TOX assays; in the case of the antiinfective screening, the throughput rises to 6400 data points per day. #Implementation Project of the Fundacion Medina at the Parque Tecnológico de Ciencias de la Salud - 25 - Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) As services, the platform offers evaluation of new compounds in: • Several drug metabolism assays (CYP P450 mediated drug-drug interactions LC-MS/MS and fluorescence assays: Inhibition P450 (CYP3A4, CYP2C9, CYP1A2, CYP2C8, CYP2C19, and CYP2D6); Inactivation and induction of CYP3A4 (CYP3A4-TDI y CYP3A4-PXR): 3500 data points/week. • Cardiotoxicity assays (ion channels: HERG, Nav1.5, Cav1.2) including binding assays, FRET, cell-based fluorescence assays, and patch clamp assays: 4000 data points/day in fluorescence assays. • Neurotoxicity assays (neurotransmitters receptors: serotonin receptors: 5HT-1A, 5HT-1B, 5HT-2A and 5HT-1C radioligand binding assays; dopamine hD2 receptor): 2000 data points/day. • Cell toxicity: MTT test in 14 tumoral and healthy cell lines: 10000 data points/day: • - Tumoral cells: (breast (MCF-7), lung (A549), liver (HepG2), colon (HT-29), ovarian (MES-SA), non resistant doxorubicin ovarian (MES-SA/Dx5), urinary bladder (HT-1197, HT-1376), skin(CRL-11147), prostate (PC-3 ) and cervix (HeLa). - Healthy cells: lung (CCD-16Lu), breast (184A1) and skin (CCD-25Sk) Infectious Diseases (including Tuberculosis and Parasitic diseases): 6400 data points/day Provision of services, facilitating the access to collections of Natural Products and the evaluation of drugs for academic research groups, biotech and pharmaceutical companies. For collaborations MEDINA offers an extensive proprietary portfolio of high-throughput anti-infective assays to facilitate lead generation (empiric whole-cell assays to evaluate the potency and spectrum of active extracts; HTS whole-cell assays with sensitized Staphylococcus aureus strains; target-based whole cell assays in Candida albicans). As MEDINA’s capabilities have expanded, so have the therapeutic screening areas, to include tuberculosis, parasitic diseases, oncology, immunoregulation and rare diseases. Our strategy is to establish scientific collaborations with academia, and biotech and pharmaceutical companies desiring to interrogate their target with one of the world’s most productive microbial extract collections. - 26 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Fundación Centro de Excelencia de Investigación en Medicamentos Innovadores en Andalucía (MEDINA) - 27 - Directory of Screening Platforms and Chemical Libraries in Spain BioLab, Instituto Universitario de Bio-Orgánica “Antonio González” Universidad de La Laguna General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 28 - Directory of Screening Platforms and Chemical Libraries in Spain BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna General Information Name and ownership BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Start-up date 2004 Contact information Name José Manuel Padrón Carrillo Postal address C/ Astrofísico Francisco Sánchez, 2 38206 La Laguna E-mail [email protected] Phone number +34 922 316 502 ext. 6126 - 29 - Directory of Screening Platforms and Chemical Libraries in Spain BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Installations Laboratory 50 m2 Usable surface area Authorization from Nuclear Safety Authority (if necessary) Office 17 m2 Other (specify) - 30 - Directory of Screening Platforms and Chemical Libraries in Spain BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Management of chemical libraries Management of chemical libraries for assays Storage capacity for compounds (number) 15.000 Storage conditions (temperature, concentration of solvents,...) -80ºC in 1.5 mL eppendorf vials Minimum volume accepted 50-500 ųl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library 40 mM solution in DMSO Coding method used in the chemical library Alphanumeric code of seven characters in the form XXX0000 State of compounds in the chemical library 40 mM solution in DMSO Number of compounds 1532 Mean molecular weight of the compounds (include standard deviation) 370 ± 148 Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products 25 % Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -20 ºC Degree of purity of compounds in the chemical library (mean value) >95% Exclusion criterion for purity (% cut-off) NO - 31 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Targets available Human Topoisomerase II alpha Types of assay Colorimetric: cytotoxicity/cell viability Flow cytometry: cell cycle disrupton, apoptosis HTS in bacterial & fungal strains: cytotoxicity/cell viability Others: E-Screen (agonist & antagonist) A-Screen (agonist & antagonist) Assay format 96 well plates Otros: 6 well plates 24 well plates 48 well plates - 32 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Growth conditions Monolayer Microorganisms Production capacity for biological reagents (nº of experimental points/week) Regularly we test 18 products in 6 cell lines, and our capacity can level up to 108 compounds per week - 33 - Directory of Screening Platforms and Chemical Libraries in Spain Infrastructure BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Description (robotized systems, detectors, etc) • At BioLab: Multiwell (96, 384) plate spectrophotometer reader. Wave lenght range 200-999 nm (BioTek PowerWave XS) • General services from ULL: Flow cytometer • Unidad Investigación HUNS Candelaria: Microarray reader - 34 - Directory of Screening Platforms and Chemical Libraries in Spain BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Personnel Fixed contract Nº % Temporary contract Function Nº % Function Doctors 3 100 Research Postgraduates 1 100 Research Technicians 1 100 Research Other personnel (specify) 1 20 Qualified (diploma level) - 35 - Graduate students Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Validation criteria per plate assayed BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Absorbance of control wells >0.2 Validation criteria per screening survey Validation criteria concentration-response curves Software for analysis of results Microsoft Excel Database for storage and consultation of results Microsoft Access/ChemFinder Accreditations and audits available - 36 - Directory of Screening Platforms and Chemical Libraries in Spain BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna Screening experience Year 2004 2005 2006 Research projects 2007 2008 (2009) 1 2 1 Contracts with industry (and size) Screening campaigns Screening conducted systematically for each batch of compounds Maximum yield per day Assays developed/optimized 108 antitumor/week; 240 antimicrobial/week Antitumor E-screen A-screen - 37 - Antimicrobial Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna As services 1. In vitro antitumor activity of synthetic or natural compounds against a panel of human solid tumor cell lines (NCI protocol) 2. Drug combination studies: analysis of synergism, antagonism and additivity (Method of Chou-Talalay) 3. Effect of the bioactive compounds on the cell cycle a) Flow cytometry (propidium iodide staining) 4. Effect of the bioactive compounds on the induction of apoptosis a) Flow cytometry (Annexin V-FITC) b) Fluorescence microscopy (DAPI) 5. (Anti)estrogenic activity in vitro (E-Screen) 6. (Anti)androgenic activity in vitro (A-Screen) 7. Antimicrobial activity of synthetic or natural compounds against a panel of bacterial a fungal strains. For collaboration Same as above - 38 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes BioLab, Instituto Universitario de Bio-Orgánica “Antonio González”, Universidad de La Laguna - 39 - Directory of Screening Platforms and Chemical Libraries in Spain APOINTECH, S.L. General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 40 - Directory of Screening Platforms and Chemical Libraries in Spain General Information APOINTECH, S.L. Name and ownership APOINTECH, S.L. Start-up date 2004 Contact information Name Mª Antonia Gómez-Zurita Frau Postal address Laboratorio 4 – CIALE C/ Río Duero Nº12 37185 – Villamayor (Salamanca) E-mail [email protected] Phone number +34 923 294500 Ext. 5105 - 41 - Directory of Screening Platforms and Chemical Libraries in Spain Installations APOINTECH, S.L. 174 m2 Usable surface area Authorization from Nuclear Safety Authority (if necessary) Other (specify) - - 42 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries APOINTECH, S.L. Management of chemical libraries for assays Storage capacity for compounds (number) >10000 Storage conditions (temperature, concentration of solvents,...) 20 ºC, liophylized or in solution (aqueous or DMSO) in a stock concentration of 0,1 or 0,01 M Minimum volume accepted 50-500 ųl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library 2 ml vials Coding method used in the chemical library Alphanumeric codes State of compounds in the chemical library Solid; Solution: Aqueous or DMSO solution, 0,10,01 M stock concentration Number of compounds 98 Mean molecular weight of the compounds (include standard deviation) Variable (generally between 350 and 600) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products 0 (all synthetic or semisynthetic molecules) Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -20ºC Degree of purity of compounds in the chemical library (mean value) >99% Exclusion criterion for purity (% cut-off) 99,00% - 43 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available APOINTECH, S.L. Targets available Receptors with enzymatic activity Kinases Transporters Others enzymes: Death receptors Caspases Apoptosis Types of assay Radiactive Luminiscence Colorimetric Fluorescence Flow cytometry HPLC/MS/MS Others: Computational genome analysis (Microarrays and High Density PCRc) Yeast deleting mutant collection Genetically modified human cells Tumor and parasitic cell variants NCI-60 panel for cancer studies Tumor cell lines from any type of tissue In vivo assays for antitumoral and antiparasitic activities Assay format 96 well plates - 44 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents APOINTECH, S.L. Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Protein production and purification Growth conditions Monolayer Suspensión Production capacity for biological reagents (nº of experimental points/week) Depending on the type of the biological reactive - 45 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure APOINTECH, S.L. Description (robotized systems, detectors, etc) • Absorbance, luminiscence and fluorescence readers. • Instrumentation and lab equipment for cell and molecular biology. • Room for biological cultivation. • Laminar flow chambers, CO2 incubators. • Flow cytometry. - 46 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel APOINTECH, S.L. Fixed contract Nº Doctors 2 % 18 Temporary contract Function Management and technical direction Nº % Function 1 9 R+D 6 55 R+D Technicians 1 9 Bioinformatics Other personnel (specify) 1 9 Laboratory technician Postgraduates Qualified (diploma level) - 47 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations APOINTECH, S.L. Validation criteria per plate assayed Triplicate assays repeated two times Validation criteria per screening survey Internal controls Validation criteria concentration-response curves Maximum and minimum values on the concentration-response curve, with a maximum of a 5% allowable error Software for analysis of results Geneprix GX and Graphpad Prism Database for storage and consultation of results Company’s own one Accreditations and audits available ISO 9001:2008 - 48 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience APOINTECH, S.L. Year 2004 2005 Research projects Contracts with industry (and size) Screening campaigns Maximum yield per day Assays developed/optimized - 49 - 2006 2007 2008 (2009) 6 6 Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered APOINTECH, S.L. As services • Screening for antitumoral agents over an extensive human tumor cells lines collection (NCI-60 and human cell lines from any type of tissue). • Screening for antiparasitic agents over an extensive parasite collection. • Mechanism of action studies for antitumoral and antiparasitic drugs. In vitro and in vivo studies. Preclinical studies. • Pharmacogenomic analisys for mechanism of action and drug resistance. For collaboration • Screening for antitumoral agents over an extensive human tumor cells lines collection (NCI-60 and human cell lines from any type of tissue). • Screening for antiparasitic agents over an extensive parasite collection. • Mechanism of action studies for antitumoral and antiparasitic drugs. In vitro and in vivo studies. Preclinical studies. We are opened to collaborations for the whole development (preclinical and clinical) of new compounds for the treatment of cancer and parasitic illnesses. - 50 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes APOINTECH, S.L. - 51 - Directory of Screening Platforms and Chemical Libraries in Spain Screening Biomar; Instituto Biomar, S.A. Translation is still pending General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 52 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Screening Biomar; Instituto Biomar, S.A. Name and ownership Screening Biomar; Instituto Biomar, S.A. Start-up date 1996 Contact information Name Antonio Fernández Medarde Postal address Instituto Biomar, S.A. Parque Tecnológico de León, Parcela M-10.4 24009 - Armunia (León) E-mail [email protected] Phone number 987 849200 - 53 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Screening Biomar; Instituto Biomar, S.A. Usable surface area 2.300 m2 Authorization from Nuclear Safety Authority (if necessary) NO Other (specify) - - 54 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries Screening Biomar; Instituto Biomar, S.A. Management of chemical libraries for assays Storage capacity for compounds (number) - Storage conditions (temperature, concentration of solvents,...) -20ºC, secos/ DMSO Minimum volume accepted 1-50 ųl Management of own chemical libraries Does the organization have its own chemical library? YES, 3 quimiotecas diferentes: extractos (BNPL-E), compuestos sin estructura asignada (BNPL-S) y compuestos con estructura asignada (BNPL-C) Format of the chemical library Tubos y placas de 96 pocillos Coding method used in the chemical library Códigos alfanuméricos State of compounds in the chemical library Sólido 40.000 extractos en la quimioteca BNPL-E; 1.500 compuestos en la quimioteca BNPL-S; 500 compuestos en la quimioteca BNPL-C Number of compounds Mean molecular weight of the compounds (include standard deviation) 487±271 (para la quimioteca BNPL-C) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products prácticamente el 100 % Does the library contain mixtures of natural products? YES Storage conditions (temperature, atmosphere, light) -20ºC secos Degree of purity of compounds in the chemical library (mean value) 75% Exclusion criterion for purity (% cut-off) - - 55 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Screening Biomar; Instituto Biomar, S.A. Targets available - Types of assay Colorimétricos Fluorescencia Assay format 96 pocillos - 56 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Screening Biomar; Instituto Biomar, S.A. Does the organization produce its own biological reagents? YES The unit has the capacity for the following Clonaje Expresión en líneas celulares Cultivo primario Growth conditions Monocapa Biorreactores Production capacity for biological reagents (nº of experimental points/week) - - 57 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Screening Biomar; Instituto Biomar, S.A. Description (robotized systems, detectors, etc) • Tecan 12 puntas – HPLC. • Detector colorimétrico – Espectrómetro De Masas. • Fluorímetro – Resonancia Magnética. - 58 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel Screening Biomar; Instituto Biomar, S.A. Fijo Nº % Función Doctors 10 - - Postgraduates 4 - - Qualified (diploma level) 1 - - Technicians 13 - - 2 - - Other personnel (specify) Contratado - 59 - Nº % Función 5 - - 5 - - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Screening Biomar; Instituto Biomar, S.A. Validation criteria per plate assayed Control positivo y negativo. Ensayos en triplicado. Validation criteria per screening survey Positivos < 2% Validation criteria concentration-response curves - Software for analysis of results - Database for storage and consultation of results Access Accreditations and audits available - - 60 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience Screening Biomar; Instituto Biomar, S.A. Año 2004 2005 2006 2007 2008 Contracts with industry (and size) 3 3 4 4 4 Screening campaigns 6 6 7 7 7 500 500 550 600 600 Research projects Maximum yield per day Assays developed/optimized - 61 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Screening Biomar; Instituto Biomar, S.A. Como servicio • Citotoxicidad = 100 compuestos/día • Antibacteriano / antifúngico = 100 compuestos/día • Angiogénesis: formación de "brotes" = 10 compuestos/día • Antioxidante = 200 compuestos/día • Blanqueador = 100 compuestos/día Como colaboración • 576 puntos de ensayo por día - 62 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Screening Biomar; Instituto Biomar, S.A. - 63 - Directory of Screening Platforms and Chemical Libraries in Spain Biomed Division – Leitat Tehnological Center General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 64 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Biomed Division – Leitat Tehnological Center Name and ownership Biomed Division – Leitat Tehnological Center Start-up date July 2008 Contact information Name Dr. Francesc Mitjans Postal address Parc Científic de Barcelona (PCB) C/ Baldiri Reixach 5-15 08028 Barcelona E-mail mailto:[email protected] Phone number +34 934020417 - 65 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Biomed Division – Leitat Tehnological Center Usable surface area 200 m2 Authorization from Nuclear Safety Authority (if necessary) - Other (specify) Access to the animal facilities and other platforms of the Scientific Park of Barcelona - 66 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries Biomed Division – Leitat Tehnological Center Management of chemical libraries for assays Storage capacity for compounds (number) 1000 Storage conditions (temperature, concentration of solvents,...) TA, -20 oC, -80 oC PBS or DMSO Minimum volume accepted 1-50 ųl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 67 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Biomed Division – Leitat Tehnological Center Targets available Peptide receptors Types of assay Luminiscence Colorimetric Fluorescence Others: In vivo models of tumor growth and metastatic dissemination both syngenic and xenogenic; as well as subcutaneous and orthotopic Assay format 96 well plates - 68 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Biomed Division – Leitat Tehnological Center Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Protein production and purification Growth conditions Monolayer Production capacity for biological reagents (nº of experimental points/week) - - 69 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Biomed Division – Leitat Tehnological Center Description (robotized systems, detectors, etc) • Infrastructure in cell culture both bacterial and tumor cell lines and endothelial cells. Models of binding, adhesion, proliferation, viability, apoptosis, migration, 3D tube formation, etc. • Luminescence, colorimetric, fluorescence readers. • Platform for generating monoclonal and polyclonal antibodies in mice. • Infrastructure of in vivo models of tumor growth and metastatic spread both syngeneic and xenogeneic; as well as subcutaneous and orthotopic. • During the first quarter-2010 we will incorporate a robotics system for ELISA and monoclonal antibody generation. - 70 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel Biomed Division – Leitat Tehnological Center Fixed contract Nº % Doctors 4 40 Postgraduates 3 40 4 40 Function Qualified (diploma level) Technicians Other personnel (specify) - 71 - Temporary contract Nº % Function Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Biomed Division – Leitat Tehnological Center Validation criteria per plate assayed - Validation criteria per screening survey - Validation criteria concentration-response curves - Software for analysis of results - Database for storage and consultation of results In evaluation phase Accreditations and audits available ISO 9001/2000 ISO 166.002/2006 - 72 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience Biomed Division – Leitat Tehnological Center Year 2004 2005 2006 2007 2008 Research projects Contracts with industry (and size) 4 SMEs Screening campaigns 2 Maximum yield per day Assays developed/optimized 4 - 73 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Biomed Division – Leitat Tehnological Center As services • Assays: cellular ELISA; cell proliferation; viability; in vitro, ex vivo and in vivo angiogenesis; in vivo models of subcutaneous and orthotopic tumors in most solid tumors both syngeneic and xenogeneic. • Currently, testing with a capacity of 200 products per week in cell-based assays • In vivo: 10 products per quarter, depending on model and test duration For collaboration After obtaining funding for collaborative projects: • Assays: cellular ELISA; cell proliferation; viability; in vitro, ex vivo and in vivo angiogenesis; in vivo models of subcutaneous and orthotopic tumors in most solid tumors both syngeneic and xenogeneic. • Currently, testing with a capacity of 200 products per week in cell-based assays • In vivo: 10 products per quarter, depending on model and test duration - 74 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Biomed Division – Leitat Tehnological Center - 75 - Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Translation is still pending General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 76 - Directory of Screening Platforms and Chemical Libraries in Spain Datos de adscripción y titularidad BIOPLATFORMSCREENTM (Thrombotargets S.L.) Name and ownership BIOPLATFORMSCREENTM propiedad de Thrombotargets SL. Start-up date Junio 2006 Contact information Name Ignasi Miquel (Director Alianzas Estratégicas) Postal address Parque Mediterráneo de la Tecnología Avenida del Canal Olímpico s/n, Edificio B6 2ª planta 08860 Castelldefels E-mail [email protected] Phone number +34936642040 - 77 - Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Instalaciones Usable surface area 80 m2 de laboratorios Authorization from Nuclear Safety Authority (if necessary) No es necesaria Other (specify) - 78 - Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Gestión de quimiotecas Management of chemical libraries for assays Storage capacity for compounds (number) 50.000 compuestos Storage conditions (temperature, concentration of solvents,...) Almacenamiento a -20ºC, disuelto en 100% DMSO Minimum volume accepted 50-500 µl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 79 - Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Ensayos disponibles Targets available Otras enzimas: Proteasas y sus moduladores Types of assay Otros: Detección física de la acción de los compuestos sin necesidad del uso de imagen Assay format 96-pocillos - 80 - Directory of Screening Platforms and Chemical Libraries in Spain Producción de reactivo biológico Does the organization produce its own biological reagents? BIOPLATFORMSCREENTM (Thrombotargets S.L.) NO The unit has the capacity for the following Growth conditions Production capacity for biological reagents (nº of experimental points/week) - 81 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructura BIOPLATFORMSCREENTM (Thrombotargets S.L.) Description (robotized systems, detectors, etc) Trombotargets dispone de laboratorios propios en su sede de Barcelona (situado en el Parque científico de Castelldefels y destinado a los departamentos de HTS y de Farmacología) y en su sede de Madrid (situado en el Centro Nacional de Biotecnología y destinado al departamento de Biología Molecular y desarrollo farmacológico). En el laboratorio de la sede de Barcelona, disponemos de dos estaciones robotizadas: • Estación 1 con una unidad robótica Hamilton con cabezal de 96 pocillos con puntas desechables (rango de 5-300µl), brazo automatizado iSWAP, dispensador de altos volúmenes (hasta 2 ml) y detector de actividad. Específica para el trabajo preparativo de las muestras (diluciones, réplicas, cherry picking etc.) y la plataforma OxidantBioplatformascreenTM. • Estación 2 con dos unidades robóticas Hamilton con cabezal de 96 pocillos con puntas desechables (rango de 5-300µl), brazo externo automatizado iSWAP, estufas para control de temperatura en MTP (rango de temperatura ambiente hasta 70ºC) y detector de actividad. Específica para el trabajo con la plataforma HemostaBioplatformscreenTM. En estos momentos, el departamento de HTS trabaja con un solo turno, ampliable a 2 turnos en caso de necesidad. - 82 - Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Personal Fijo Nº % Función Contratado Nº Doctors Postgraduates 1 100 Responsable de la unidad Qualified (diploma level) 1 100 Trabajo de laboratorio Technicians 1 100 Trabajo de laboratorio Other personnel (specify) - 83 - % Función Directory of Screening Platforms and Chemical Libraries in Spain Control de calidad y acreditaciones BIOPLATFORMSCREENTM (Thrombotargets S.L.) Validation criteria per plate assayed Controles positivos y negativos del ensayo en cada placa analizada Validation criteria per screening survey Controles rutinarios diarios de la plataforma y de verificación semanal de los equipos Validation criteria concentration-response curves Ensayos complementarios a la plataforma y uso de inhibidores de la respuesta Software for analysis of results Software propio Database for storage and consultation of results Estructura propia en nuestro servidor Accreditations and audits available • - 84 - Aunque hasta el momento no disponemos de acreditaciones ni auditorías que certifiquen nuestras plataformas, en Thrombotargets trabajamos en condiciones que se corresponden con las directrices de las Buenas Prácticas de Laboratorio. Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Experiencia en Screening Año 2004 2005 2006 2007 2008 Research projects 2 3 2 Contracts with industry (and size) 5 3 3 Screening campaigns 1 1 1 400 4000 8000 1 1 1 Maximum yield per day Assays developed/optimized - 85 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Capacidades disponibles para ofertar BIOPLATFORMSCREENTM (Thrombotargets S.L.) Como servicio • HemostaBioplatformscreenTM: Plataforma destinada a la identificación de compuestos con potenciales actividades de procoagulación, anticoagulación, profibrinolísis y antifibrinolísis. Esta plataforma es un ensayo relacionado la vía de la Hemostasia completa pudiendo evaluar cualquier paso de la cascada de la coagulación. Capacidad: 6400 compuestos/día por turno de 8 horas. Como colaboración • OxidantBioplatformscreenTM: Plataforma destinada a la identificación de compuestos con potenciales actividades de antioxidación plasmática total. Esta plataforma es específica para el proceso oxidativo asociado con enfermedades neurodegenerativas y envejecimiento. Capacidad: 1600 compuestos/día por turno de 8 horas. • AtheroBioplatformscreenTM: Plataforma destinada a la identificación del efecto antiaterosclerótico de los compuestos analizados mediante la inhibición de la modificación de lipoproteinas de baja densidad por lipoperoxidación. Capacidad: 20 compuestos/día por turno de 8 horas. - 86 - Directory of Screening Platforms and Chemical Libraries in Spain BIOPLATFORMSCREENTM (Thrombotargets S.L.) Anexos Resumen de ventajas. Adicionalmente, existe un folleto describiendo en detalle BIOPLATFORMSCREEN que se puede obtener de la dirección de contacto. - 87 - Directory of Screening Platforms and Chemical Libraries in Spain NEUROSCIENCE TECHNOLOGIES S.L.P. Translation is still pending General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 88 - Directory of Screening Platforms and Chemical Libraries in Spain Datos de adscripción y titularidad NEUROSCIENCE TECHNOLOGIES S.L.P. Name and ownership NEUROSCIENCE TECHNOLOGIES S.L., titularidad privada Start-up date 3/6/2005 Contact information Name Cristina Quiles Postal address Parc Científic de Barcelona Edificio Hélix, Baldiri Reixac 15-21 08028 Barcelona E-mail [email protected] Phone number 93 4020164 - 89 - Directory of Screening Platforms and Chemical Libraries in Spain Instalaciones NEUROSCIENCE TECHNOLOGIES S.L.P. 120 m2 Usable surface area Authorization from Nuclear Safety Authority (if necessary) Other (specify) - 90 - Directory of Screening Platforms and Chemical Libraries in Spain Gestión de quimiotecas NEUROSCIENCE TECHNOLOGIES S.L.P Management of chemical libraries for assays Storage capacity for compounds (number) 10.000 Storage conditions (temperature, concentration of solvents,...) Temperatura ambiente, 4ºC, -20ºC, -80ºC, nitrógeno líquido Minimum volume accepted 5 µl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 91 - Directory of Screening Platforms and Chemical Libraries in Spain Ensayos disponibles NEUROSCIENCE TECHNOLOGIES S.L.P. Targets available Canales iónicos tanto voltaje-dependientes como dependientes de ligandos. Fibras nerviosas del Sistema nervioso Periférico Types of assay a) Ensayos farmacológicos en canales iónicos (patch clamp automatizado) a. Concentraciones puntuales b. Curvas concentración-respuesta b) Electromiografia y estudios de conducción nerviosa c) Potenciales evocados a. Potenciales evocados sensoriales y motores b. Potenciales evocados por láser c. Potenciales evocados por calor de contacto (CHEPS) d) Microneurografía a. Estudios de transducción sensorial b. Índice de ectopia (NT-ectopia) e) Estudios de excitabilidad nerviosa (MEM) a. Curva de estímulo-respuesta b. Threshold electrotonus c. Relación corriente/voltaje d. Recovery cycle f) Immunohistoquímica de fibras nerviosas intraepidérmicas a. Cuantificación de fibras nerviosas g) Estudios de comportamiento animal a. Test plantar de Hargreaves b. Estesiómetro plantar dinámico Assay format Ensayos en Patch clamp: Placas de 96 pocillos o en viales de vidrio de 2ml. Resto de ensayos: Viales de vidrio de 2ml - 92 - Directory of Screening Platforms and Chemical Libraries in Spain Producción de reactivo biológico Does the organization produce its own biological reagents? The unit has the capacity for the following Growth conditions NEUROSCIENCE TECHNOLOGIES S.L.P. YES Expresión de canales iónicos en líneas celulares Cultivos primarios y de líneas celulares. Monocapa Production capacity for biological reagents (nº of experimental points/week) - 93 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructura NEUROSCIENCE TECHNOLOGIES S.L.P. Description (robotized systems, detectors, etc) • Unidad de Patch-clamp automatizado: 3 canales de registro simultáneo • Unidad de cultivos celulares • Unidad de histología • Unidad de electrofisiología in vivo: 4 equipos de registro simultáneo • Unidad de comportamiento animal: 2 equipos - 94 - Directory of Screening Platforms and Chemical Libraries in Spain Personal NEUROSCIENCE TECHNOLOGIES S.L.P. Fijo Nº % Función Doctors 3 100 Investigadores básicos Postgraduates 5 100 Investigadores clínicos, personal administrativo Qualified (diploma level) Technicians Other personnel (specify) - 95 - Contratado Nº % Función 3 25 Investigadores clínicos Directory of Screening Platforms and Chemical Libraries in Spain Control de calidad y acreditaciones NEUROSCIENCE TECHNOLOGIES S.L.P. Validation criteria per plate assayed Porcentajes de inhibición de la corriente iónica para cada compuesto. Validation criteria per screening survey Se comparan con los obtenidos en fármacos control. Porcentajes de inhibición de la corriente iónica para cada compuesto. Validation criteria concentration-response curves Ajuste de los valores obtenidos a una curva sigmoidea. Análisis estadístico de la misma. Los valores se comparan con los obtenidos en fármacos control Software for analysis of results QTRAC, FLYSCREEN SUITE DATA; PATCHMASTER Database for storage and consultation of results EXCEL, ACCESS Accreditations and audits available En proceso - 96 - Directory of Screening Platforms and Chemical Libraries in Spain Experiencia en Screening NEUROSCIENCE TECHNOLOGIES S.L.P. Año 2004 2005 2006 2007 2008 Research projects Contracts with industry (and size) (2009) 4 1 Screening campaigns Maximum yield per day Assays developed/optimized - 97 - 2 2 3 Directory of Screening Platforms and Chemical Libraries in Spain Capacidades disponibles para ofertar NEUROSCIENCE TECHNOLOGIES S.L.P. Como servicio • Ensayos farmacológicos en canales iónicos (PATCH CLAMP): 30 data points/día • Electromiografía: 5 data points/día • Electroneurografía 5 data points/día • Microneurografía: 4 data points/día • Immunohistoquímica: 4 data points/semana • Estudios de comportamiento animal: 4 data points/día Como colaboración • Ensayos farmacológicos en canales iónicos (PATCH CLAMP): 30 data points/día • Electromiografía: 5 data points/día • Electroneurografía 5 data points/día • Microneurografía: 4 data points/día • Immunohistoquímica: 4 data points/semana • Estudios de comportamiento animal: 4 data points/día - 98 - Directory of Screening Platforms and Chemical Libraries in Spain Anexos NEUROSCIENCE TECHNOLOGIES S.L.P. - 99 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 100 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) General Information Name and ownership USEF Screening Platform Start-up date 2004 Contact information Name Mabel Loza Postal address School of Pharmacy Pharmacology Department E-15782 Santiago de Compostela. Spain E-mail [email protected] Phone number +34 981 563 100 ext: 15005 - 101 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Installations Usable surface area 250 m2 Authorization from Nuclear Safety Authority (if necessary) YES Other (specify) - - 102 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Management of chemical libraries Management of chemical libraries for assays Storage capacity for compounds (number) 20.000 cp Storage conditions (temperature, concentration of solvents,...) -20º, -80º y 4º Minimum volume accepted 1-50ųl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library 96 well plates Coding method used in the chemical library Alphanumeric code/ bar code State of compounds in the chemical library Solid Solution Number of compounds 12.000 cp Mean molecular weight of the compounds (include standard deviation) 400 ± 150 Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products 0,00% Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -20 ºC, -80 ºC and 4 ºC Light protected Degree of purity of compounds in the chemical library (mean value) 90,00% Exclusion criterion for purity (% cut-off) <80% - 103 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Assays available Targets available GPCRs Ion channels Peptide receptors Kinases Phosphatases Transporters Other enzymes: Phosphodiesterases Types of assay Radiactive: Filtrate SPA Luminiscence Colorimetric Fluorescence Polarised fluorescence TRF FRET BRET Assay format 96 well plates 384 well plates - 104 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Growth conditions Monolayer Spinner flasks Production capacity for biological reagents (nº of experimental points/week) 5.000 experimental points/week - 105 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Description (robotized systems, detectors, etc) • A modern and well equipped laboratory is available for assessment of the pharmacological activity of molecules, by a team of specialized professionals capable of evaluating the different aspects of planning screening cascades and target validation for drug discovery. • The infrastructures required for carrying out large scale drug screening cascades, for both binding and functional studies, are available. These include a Freedom Evo robot (Tecan) for handling liquids, and an Aquarius robot (Tecan) for handling 96- and 384-well plates. • Multidrop systems (TermoLab Systems), equipped with a Titan Stacker robotic arm (ThermoLab Systems) are available for addition of liquids to 96- and 384- well plates, and to deep well plates. A Columbus plate washer (Tecan) enables maximum yields in assays that require excess colour to be washed from plates (cytotoxicity, immunoenzyme assays). • A multifunctional Ultra Evolution detector (Tecan) is also available, enabling colorimetric, fluorescence, polarized fluorescence, FRET and luminescence determinations; the detector is equipped with a Connect robotic arm (Tecan), which enables the simultaneous automated detection of up to 16, 96-well plates or 32, 384-well plates. A Microbeta Trilux detector (Wallac) is also available • A fluorescence microscope coupled to dual detection and rapid image acquisition systems is also available for temporal and spatial resolution of cell events, combined with image analysis software, which enables highly sensitive and selective cell culture assays to be carried out. • A modern system of thermostat controlled water baths is also available for “in vitro” and “ex vivo” functional studies with a simultaneous series of transducers and polygraphs (Grass). - 106 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Personnel Fixed contract Nº % Temporary contract Function Nº % Function 2 Area responsible person (HTS and biological reagents) Postgraduates 1 Laboratory Qualified (diploma level) 1 Management Technicians 6 Laboratory Other personnel (specify) 1 Informatics Doctors 2 Directorship - 107 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Validation criteria per plate assayed Z’ scores, signal-noise ratio, reference commercial compounds Validation criteria per screening survey Z’ scores distribution, signal-noise ratio distribution and reference commercial compounds distribution Validation criteria concentration-response curves Curve slope, signal-noise ratio, reference commercial compounds affinity Software for analysis of results Xlfit Database for storage and consultation of results Own Accreditations and audits available - - 108 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) Screening experience Year 2004 2005 Research projects Contracts with industry (and size) 3 4 Screening campaigns 2006 2007 2008 1 5 8 4 5 9 25 Maximum yield per day 1800 Assays developed/optimized 12 - 109 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) As services • Radioligand binding assays: 1800 cp/day • Enzimatic assays: 3600 cp/day • Second messenger measurement assays: 360 cp/day • Cytochrome inhibition assays: 360 cp/day • Cytotoxicity assays: 30 cp/day • Isolated organ assays: 15 cp/day For collaboration The same as above - 110 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Plataforma de Screening de Fármacos USEF (Universidad de Santiago de Compostela) - 111 - Directory of Screening Platforms and Chemical Libraries in Spain NEUROLAB_IQM General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 112 - Directory of Screening Platforms and Chemical Libraries in Spain General Information NEUROLAB_IQM Name and ownership NEUROLAB_IQM Start-up date 2004 Contact information Name Mª Isabel Rodríguez Franco / Ana Martínez Gil Postal address Instituto Química Médica (CSIC) C/ Juan de la Cierva, 3 28006-Madrid E-mail [email protected] [email protected] Phone number +34 91 5680010 / 912587482 - 113 - Directory of Screening Platforms and Chemical Libraries in Spain Installations NEUROLAB_IQM Usable surface area 15 m2 Authorization from Nuclear Safety Authority (if necessary) N/A Other (specify) - - 114 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries NEUROLAB_IQM Management of chemical libraries for assays Storage capacity for compounds (number) 10 a 100 Storage conditions (temperature, concentration of solvents,...) 25 ºC, solid (preferably) Minimum volume accepted Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library Tubes Coding method used in the chemical library Alphanumerical codes State of compounds in the chemical library Solid Number of compounds >1000 Mean molecular weight of the compounds (include standard deviation) 200-500 Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products 0% Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Room temperature, darkness Degree of purity of compounds in the chemical library (mean value) 98% Exclusion criterion for purity (% cut-off) >95% - 115 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available NEUROLAB_IQM Targets available Other assays: BBB-PAMPA (CNS Penetration with Parallel Artificial Membrane Permeation Assay) AO-PAMPA (Oral Absortion with Parallel Artificial Membrane Permeation Assay) Types of assay Colorimétricos Fluorescence Assay format 96 well plates - 116 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Does the organization produce its own biological reagents? NEUROLAB_IQM NO The unit has the capacity for the following Growth conditions Production capacity for biological reagents (nº of experimental points/week) - 117 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure NEUROLAB_IQM Description (robotized systems, detectors, etc) • UV / Vis microplate (96- and 384-wells) and cuvette spectrophotometer - 118 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel NEUROLAB_IQM Fixed contract Nº % Temporary contract Function Doctors Nº 2 Postgraduates Qualified (diploma level) Technicians Other personnel (specify) - 119 - % 20 Function PAMPA assays Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations NEUROLAB_IQM Validation criteria per plate assayed 10-15 commercial drugs of known properties Validation criteria per screening survey - Validation criteria concentration-response curves N/A Software for analysis of results - Database for storage and consultation of results - Accreditations and audits available - - 120 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience NEUROLAB_IQM Year 2004 2005 Research projects 2006 2007 1 1 2008 (2009) Contracts with industry (and size) 1 1 Screening campaigns 2 1 2 3 3 4 Maximum yield per day 5 5 10 10 10 10 Assays developed/optimized 1 1 2 2 2 2 - 121 - Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered NEUROLAB_IQM As service • In vitro determination of the oral absorption and central nervous system penetration, using PAMPA assays For collaboration • In vitro determination of the oral absorption and central nervous system penetration, using PAMPA assays. - 122 - Directory of Screening Platforms and Chemical Libraries in Spain Anexos NEUROLAB_IQM Additional information PAMPA assays have been included in the following papers and patents, authored by the NEUROLAB_IQM group: 1. Rodríguez-Franco, M. I.; Fernández-Bachiller, M. I.; Pérez, C.; Hernández-Ledesma, B.; Bartolomé, B. Novel tacrine – melatonin hybrids as dual-acting drugs for Alzheimer disease, with improved acetylcholinesterase inhibitory and antioxidant properties. J. Med. Chem. 2006, 49, 459-462. 2. Pavón, F. J.; Hernández-Folgado, L.; Bilbao, A.; Cippitelli, A.; Jagerovic, N.; Abellán, G.; Rodríguez-Franco, M. I.; Serrano, A.; Macías, M.; Navarro, M.; Goya, P.; Rodríguez de Fonseca, F. Antiobesity effects of the novel in vivo neutral cannabinoid receptor antagonist 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-3-hexyl-1H-1,2,4-triazole - LH 21. Neuropharmacology 2006, 51, 358-366. 3. Reviriego, F.; Rodríguez-Franco, M. I.; Navarro, P.; García-España, E.; Liu-González, M.; Verdejo, B.; Domènech, A. The sodium salt of diethyl 1H-pyrazole-3,5-dicarboxylate as an efficient amphiphilic receptor for dopamine and amphetamines. Crystal structure and solution studies. J. Am. Chem. Soc. 2006, 128, 16458-16459. 4. Fernández-Bachiller, M. I.; Pérez, C.; Campillo, N. E.; Páez, J. A.; González-Muñoz, G. C.; Usán, P.; García-Palomero, E.; López, M. G.; Villarroya, M.; García, A. G.; Martínez, A.; Rodríguez-Franco, M. I. Tacrine – Melatonin Hybrids as Multifunctional Agents for Alzheimer’s Disease, with Cholinergic, Antioxidant, and Neuroprotective Properties. ChemMedChem 2009, 4, 828-841. 5. Marco-Contelles, J.; León, R.; de los Ríos, C.; Samadi, A.; Bartolini, M.; Andrisano, V.; Huertas, O.; Barril, X.; Luque, F. J.; Rodríguez-Franco, M. I.; López, B.; López, M. G.; García, A. G.; Carreiras, M. C.; Villarroya, M. Tacripyrines, the first tacrine-dihydropyridine hybrids, as multitarget-directed ligands for the treatment of Alzheimer’s disease. J. Med. Chem. 2009, 52, 2724-2732. 6. Camps, P.; Formosa, X.; Galdeano, C.; Muñoz-Torrero, D.; Ramírez, L.; Gómez, E.; Isambert, N.; Lavilla, R.; Badia, A.; Clos, M. V.; Bartolini, M.; Mancini, F.; Andrisano, V.; Arce, M. P.; Rodríguez-Franco, M. I.; Huertas, O.; Dafni, T. and Luque, F. J. Pyrano[3,2-c]quinoline-6-Chlorotacrine Hybrids as a Novel Family of Acetylcholinesterase- and β-Amyloid-Directed Anti-Alzheimer Compounds. J. Med. Chem. 2009, 52, 5365–5379. 7. Arce, M. P.; Rodríguez-Franco, M. I.; González-Muñoz, G. C.; Pérez, C.; López, B.; Villarroya, M.; López, M. G.; García, A. G.; Conde, S. Neuroprotective and Cholinergic Properties of Multifunctional Glutamic Acid Derivatives for the Treatment of Alzheimer’s Disease. J. Med. Chem. 2009, 52, 7249-7257. 8. Conde, S.; Rodríguez-Franco, M. I.; Arce, M. P.; González-Muñoz, G. C.; Villarroya, M.; López, M. G.; García, A. G. Novel dicarboxylic amino acid derivatives and the use thereof in the treatment of neurodegenerative diseases. ES Pat. P200703264; PCT/ES2008/070221;.WO 2009/074706 A1. 9. Conde, S.; Rodríguez-Franco, M. I.; González-Muñoz, G. C.; Arce, M. P.; Villarroya, M.; López, M. G.; García, A. G. Use of heterocyclic hydrazides in the treatment of neurodegenerative diseases. ES Pat. P200801900; PCT/ES2009/070229. 10. Conde, S.; Rodríguez-Franco, M. I.; González-Muñoz, G. C.; Villarroya, M.; López, M. G.; García, A. G. 1,4,5-Dibenzo[b,f]thiadiazepines 5,6-dihydro as neuroprotective agents, useful for the treatment of Alzheimer’s and other neurodegenerative diseases. ES Pat. 200803413. PCT/ES2009/070522. 11. Rodríguez-Franco, M. I.; Conde, S.; López-Iglesias, B.; Pérez, C.; Villarroya, M.; López, M. G.; García, A. G. New derivatives of bis(aralkyl)amino and 6-membered (hetero)aromatic systems and the use thereof in the treatment of neurodegenerative diseases, including Alzheimer’s disease. ES Pat. 200930931. 12 Rodríguez-Franco, M. I.; Conde, S.; López-Iglesias, B.; Pérez, C.; Villarroya, M.; López, M. G.; García, A. G. New derivatives of bis(aralkyl)amino and [6+5]-heteroaromatic systems and the use thereof in the treatment of neurodegenerative diseases, including Alzheimer’s disease. ES Pat. 200930936. - 123 - Directory of Screening Platforms and Chemical Libraries in Spain MEJORAN LAB AUTOMATION S.L. General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 124 - Directory of Screening Platforms and Chemical Libraries in Spain General Information MEJORAN LAB AUTOMATION S.L. Name and ownership MEJORAN LAB AUTOMATION S.L. Start-up date 2001 Contact information Name Juan Bautista Crespo García Postal address MEJORAN LAB AUTOMATION S.L. c/ Lope de Rueda 3, 1º Dcha. 28008 Madrid E-mail [email protected] Phone number 655894876 - 125 - Directory of Screening Platforms and Chemical Libraries in Spain Installations MEJORAN LAB AUTOMATION S.L. Usable surface area 200 m2 Authorization from Nuclear Safety Authority (if necessary) - Other (specify) - - 126 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries MEJORAN LAB AUTOMATION S.L. Management of chemical libraries for assays Storage capacity for compounds (number) 7000 Storage conditions (temperature, concentration of solvents,...) -80º C Minimum volume accepted 50-500 µl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library 96-well plates; IN SILICO Coding method used in the chemical library - State of compounds in the chemical library Solution Number of compounds 7000 (planned) Mean molecular weight of the compounds (include standard deviation) 3000 Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products 30-40 Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) - Degree of purity of compounds in the chemical library (mean value) - Exclusion criterion for purity (% cut-off) - - 127 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available MEJORAN LAB AUTOMATION S.L. Targets available GPCRs Ion channels Kinases Phosphatases Transporters Types of assay Luminiscence Colorimetric Fluorescence Automated patch-clamp Flow cytometry Zebra fish HTS in bacterial strains Steam cells differentiation Assay format 96-well plates - 128 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents MEJORAN LAB AUTOMATION S.L. Does the organization produce its own biological reagents? YES The unit has the capacity for the following - Growth conditions - Production capacity for biological reagents - (nº of experimental points/week) - 129 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure MEJORAN LAB AUTOMATION S.L. Description (robotized systems, detectors, etc) • Liquid Handlers: TECAN GENESIS(1), THERMO MULTIDROP(1), BIOTEK PRECISIONM(1), TOMTEC QUADRA(4) • Detectors: TECAN SPECTRAFLUOR(2) SAFIRE(1). • Others: CENTRIFUGAS (Ultra y Micro), AGITADORES, BALANZAS, LAVADORES, STACKERS, CONGELADORES, ETC. - 130 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel MEJORAN LAB AUTOMATION S.L. Fixed contract Nº % Doctors 1 10 Postgraduates 4 40 Qualified (diploma level) 3 30 Function MANAGER LAB AUTOM & BIOINFORMATICA Desarrolladores de software y administración Technicians Other personnel (specify) - 131 - Temporary contract Nº 2 3 3 % Function Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations MEJORAN LAB AUTOMATION S.L. Validation criteria per plate assayed - Validation criteria per screening survey - Validation criteria concentration-response curves - Software for analysis of results Nautilus LIMS Database for storage and consultation of results Oracle Accreditations and audits available ISO 9001:200 - 4001:2004 - 132 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience MEJORAN LAB AUTOMATION S.L. Year 2004 Research projects Contracts with industry (and size) Screening campaigns Maximum yield per day Assays developed/optimized - 133 - 2005 2006 2007 2008 (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered MEJORAN LAB AUTOMATION S.L. As services • Virtual Screening • In Vitro and Cell-based bacterial and mamalian cells • Colorimetric, fluorimetric and luminiscense based screens For collaboration • 500 points/week in vitro and living cells • Virtual Screening 4.5 x 106 /week - 134 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes MEJORAN LAB AUTOMATION S.L. - 135 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 136 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN General Information Name and ownership Screening of physical interactions based on NMR (1H and 19F) of fluorinated fragments Spectroscopy and NMR Unit Spanish National Cancer Center Start-up date 01/08/08 Contact information Name Ramón Campos Olivas Postal address C. Melchor Fernández Almagro, 3 Madrid 28029 E-mail [email protected] Phone number +34 917328000 - 137 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Installations Usable surface area 30 m2 Authorization from Nuclear Safety Authority (if necessary) N/A Other (specify) - - 138 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Management of chemical libraries Management of chemical libraries for assays Storage capacity for compounds (number) YES, near 2.000 Storage conditions (temperature, concentration of solvents,...) At -20ºC, as powder or stock solutions in d6-DMSO Minimum volume accepted 1-50 ųl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library Tubes Coding method used in the chemical library Manual annotation and grouping in cocktails of 8 cmpds State of compounds in the chemical library Solid Solution: 80 mM in d6-DMSO Number of compounds 380 Mean molecular weight of the compounds (include standard deviation) 265 (55) Characteristics of the chemical library Average number of H donors and acceptors #donnors: 3.8(1.5); #acceptors: 1.3(0.9) Percentage of natural products 0 Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -20 ºC Degree of purity of compounds in the chemical library (mean value) > 95% Exclusion criterion for purity (% cut-off) - - 139 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Assays available Targets available Types of assay Fluorescence Polarised fluorescence Others: Nuclear Magnetic Resonance (NMR) for detection of changes in the small molecule signals upon receptor binding. No restrictions on the type or size of the receptor, if it can be solubilized to ~1-0.1 micromolar concentration. Assay format Other: The fluorinated compounds of our chemical library have been grouped in mixtures of 8 compounds each. The assignment of the 19F NMR signals from each mixture is available and the identification of the component (putative ligand) being affected by adding the receptor to a mixture is immediate. The experiments are done in 5mm NMR tubes containing 20-50 microM for each compounds and o.1-1 micromolar receptor, in a volume of 500-600microL, using the appropriate buffer for the receptor. We have a sample exchanger with capability to measure up to 120 samples without human intervention. - 140 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Production of biological reagents Does the organization produce its own biological reagents? YES The unit has the capacity for the following Protein production and purification Growth conditions Bacterial over-expression in shaking flasks Production capacity for biological reagents (nº of experimental points/week) - - 141 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Description (robotized systems, detectors, etc) • NMR spectrometer of 700 MHz (Bruker Avance) which is well equipped with probes (HR-MAS, dual fluorine/proton, and triple and quadruple resonance) • The NMR spectrometer is equipped with a sample changer (Bruker BACS120) that is able to run up to 120 samples without human intervention, providing the required throughput for the NMR-based screening - 142 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Personnel Fixed contract Nº Doctors 2 % Function 100 Postgraduates Qualified (diploma level) Technicians Other personnel (specify) - 143 - Temporary contract Nº % Function Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Validation criteria per plate assayed Reproducibility of negative, not-affected compounds in the mixture Validation criteria per screening survey Individual examination of the hit compound at different concentrations of hit and receptor, and using alternative 1H-NMR methods Validation criteria concentration-response curves NO Software for analysis of results Topspin(Bruker) Database for storage and consultation of results Not available, manual annotation Accreditations and audits available NO - 144 - Directory of Screening Platforms and Chemical Libraries in Spain Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN Screening experience Year 2004 2005 2006 2007 2008 Research projects Contracts with industry (and size) Screening campaigns 3 Maximum yield per day Assays developed/optimized 4 - 145 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN As services • Screening of the fluorinated fragment library (380 cmpds) using 19F-NMR to identify possible ligands (Phase I, mixture evaluation): 3 days. • Hit verification using 19F and 1H NMR methods at different concentrations (Phase II, individual examination of Phase I hits): 0.5 days/hit. For collaboration • Screening of the fluorinated fragment library (380 cmpds) using 19F-NMR to identify possible ligands (Phase I, mixture evaluation): 3 days. • Hit verification using 19F and 1H NMR methods at different concentrations (Phase II, individual examination of Phase I hits): 0.5 days/hit. - 146 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Centro Nacional de Investigaciones Oncológicas (CNIO). Unidad de Espectroscopía y RMN - 147 - Directory of Screening Platforms and Chemical Libraries in Spain Vivia Biotech SL General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 148 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Vivia Biotech SL Name and ownership Vivia Biotech SL Start-up date 25 Julio 2007 Contact information Name Joan Ballesteros Postal address C/ Moratín 50, 3-Dcha 28014 Madrid E-mail [email protected] Phone number 649872032 - 149 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Vivia Biotech SL Usable surface area 400 m2 Authorization from Nuclear Safety Authority (if necessary) NO Other (specify) - - 150 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries Vivia Biotech SL Management of chemical libraries for assays Storage capacity for compounds (number) 500.000 Storage conditions (temperature, concentration of solvents,...) DMSO solutions, 10mM. -20º C. Minimum volume accepted 1-50 ųl Management of own chemical libraries Does the organization have its own chemical library? YES Format of the chemical library 96-well plates Coding method used in the chemical library Bar codes labels (associated to ActivityBase database with associated screening data) State of compounds in the chemical library Solution: DMSO 10mM Number of compounds 2840 Mean molecular weight of the compounds (include standard deviation) 365,0 ± 177,5 Characteristics of the chemical library Average number of H donors and acceptors 3,6 Percentage of natural products 10% Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) Freezers -20ºC Degree of purity of compounds in the chemical library (mean value) 97% Exclusion criterion for purity (% cut-off) 90% - 151 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Vivia Biotech SL Targets available GPCRs Other enzymes: Phenotypic assays (apoptosis, oxidative stress) using cell lines and whole blood. Types of assay Fluorescence Flow cytometry Assay format 96 well plates - 152 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Vivia Biotech SL Does the organization produce its own biological reagents? NO The unit has the capacity for the following Cloning Cell line expression Primary cultures Growth conditions Monolayer Spinner flasks Production capacity for biological reagents (nº of experimental points/week) - - 153 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Vivia Biotech SL Description (robotized systems, detectors, etc) 4 integrated platforms of screening by flow citometry “ExviTech” consisting of: Automated liquid handlers (Biomek 3000) inside laminar flow hoods. Flow citometers Beckman-Coulter: Cyan 3 lasers (3 units) and 1 sorter FACS MoFlo 3 lasers. 4 Auto-samplers for flow citometry using custom proprietary technology. Screening software and database: ActivityBase from IDBS and custom analysis software. - 154 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel Vivia Biotech SL Fixed contract Temporary contract Nº % Function Nº % Function Doctors 9 100 Scientific 2 50 Project managers Postgraduates 8 100 Scientific 5 100 Administration Qualified (diploma level) Technicians Other personnel (specify) - 155 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Vivia Biotech SL Validation criteria per plate assayed Z’ ≥ 0.4 ; SW≥2 (Signal Window) Validation criteria per screening survey Controls and standards variability lower than 10% with respect to historical values. Validation criteria concentration-response curves 4P logistic non-lineal curve fit model. Software for analysis of results XE Runner/Xlfit ActivityBase IDBS Database for storage and consultation of results Activitybase IDBS Accreditations and audits available - - 156 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience Vivia Biotech SL Year 2004 2005 2006 2007 2008 4000 4000 4000 1000 2000 20 35 52 55 62 Research projects Contracts with industry (and size) Screening campaigns Maximum yield per day Assays developed/optimized - 157 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Vivia Biotech SL As services • 52 GPCRs Calcium coupled expressed in cell lines. • Screening GPCR allosteric modulators: 2000/day in 5 GPCRs in parallel. • Standard screening: 2000 per day in 5-10 GPCRs in parallel. For collaboration Same as services and only as collaboration: • Screening of 2000 compounds in blood samples from patients either peripheral blood or bone marrow. • Apoptosis evaluation in malignant hematologies, or oxidative stress levels. Other additional parameters under development. - 158 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Vivia Biotech SL - 159 - Directory of Screening Platforms and Chemical Libraries in Spain BIOBIDE S.L. General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 160 - Directory of Screening Platforms and Chemical Libraries in Spain General Information BIOBIDE S.L. Name and ownership BIOBIDE, S.L. Start-up date December 2005 Contact information Name Arantza Muriana Postal address Paseo Mikeletegi 58 San Sebastián 2012 E-mail [email protected] Phone number +34 943309360 - 161 - Directory of Screening Platforms and Chemical Libraries in Spain Installations BIOBIDE S.L. Usable surface area 1.000 m2 Authorization from Nuclear Safety Authority Not applicable (if necessary) Other (specify) Permission to work with experimental animals and GMO's, and permission of Industry. Also registered agricultural center. - 162 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries BIOBIDE S.L. Management of chemical libraries for assays Storage capacity for compounds (number) > 1.000 Storage conditions (temperature, concentration of solvents,...) -80ºC, 20ºC y 4ºC, also solve in different solvents (usually DMSO) Minimum volume accepted 50-500 µl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 163 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available BIOBIDE S.L. Targets available Types of assay Fluorescence Zebra fish Assay format 96-well plates 24-well plates - 164 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents BIOBIDE S.L. Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary culture Growth conditions Massively crossing of adult zebrafish for embryos production or individual crossing of pairs of adults to obtain embryos of the same line for lines maintenance. Production capacity for biological reagents (nº of experimental points/week) 4.800 zebrafish embryos / day - 165 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure BIOBIDE S.L. Description (robotized systems, detectors, etc) • HTS Platform for automated zebrafish embryos. • LIMS and image analysis software (photo and video) developed in house. • Equipments for zebrafish microinjection, and magnifying glasses. • Microscope, stereomicroscope, incubators, liquid handling, dispenser of embryos, and so on. • Facility and installations of zebrafish for fish keeping with automated water control parameters system (software) and alarms. - 166 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel BIOBIDE S.L. Fixed contract Nº % Temporary contract Function Nº % Project Leaders and Quality Director HTS, Facility and R&D responsibles, as well as Executive and Administrative Directors Doctors 5 30 Postgraduates 9 82 Qualified (diploma level) 1 5.9 HTS Technitian Technicians 2 11.8 Zebrafish facility Technitian Other personnel (specify) - 167 - 1 5.9 1-2 5.9 Function Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations BIOBIDE S.L. Validation criteria per plate assayed Minimum number of images obtained per plate, a negative control per plate and a minimum distance between test population and control population Validation criteria per screening survey Applicable acceptance criteria of plates to all the plates that make up a screening campaign Validation criteria concentration-response curves Applicable acceptance criteria of plates to all the plates needed to make a concentration-response curve Software for analysis of results Image analysis software (photo and video) for each type of test, developed in house; statistical software (descriptive and others). Database for storage and consultation of results Databases hosted on multiple servers to maintain the image of testing and databases management LIMS with guaranteed traceability. Accreditations and audits available Obtained certification of Good Laboratory Practice (GLP's) in August 2009 with scope for in vivo toxicity, pharmacodynamics and safety pharmacology. Internal audit commissioned by an external quality consultant at the University of Navarra (CIFA) previous to GLP's audit of the Spanish Agency of Medicines (February '09) - 168 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience BIOBIDE S.L. Year 2004 2005 Research projects 2006 2007 2008 (2009) 6 9 Contracts with industry (and size) 6 12 Screening campaigns NA NA Maximum yield per day 540 540 4 Assays developed/optimized 5 1 - 169 - 2 6 Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered BIOBIDE S.L. As services • Cardiotox assay- cardiotoxicity test for predicting potential QT prolongation: 6- in zebrafish: 36 compounds per day* • Teratox assay: Test of Teragenicity in zebrafish: 2-4 compounds / day • Acutetox assay: general toxicity test in zebrafish: 2-4 compounds / day • Ototoxic assay: test of ototoxicity in zebrafish: 2-6 compounds / day • Angiogenesis assay: Test of inhibition / activation of angiogenesis for regenerative or efficacy of anticancerous drugs: with 2-10 compounds per day • Alzheimer's efficacy: acetylcholinesterase inhibition test: 2-6 compounds / day (*) The number of compounds per day depends on whether the test is done at 1 or 2 Concentracions, or with concentration-response curves of more than 5 points For collaboration • Cardiotox assay- cardiotoxicity test for predicting potential QT prolongation: 6- in zebrafish :36 compounds per day • Teratox assay: Test of Teragenicity in zebrafish: 2-4 compounds / day • Acutetox assay: general toxicity test in zebrafish: 2-4 compounds / day • Ototoxic assay: test of ototoxicity in zebrafish: 2-6 compounds / day • Hepatotox assay: Test of hepatotoxicity in zebrafish: 6 compounds per day • Neurotox assays: Tests of neurotoxicity in zebrafish: 6 compounds per day • Effectiveness of Anticancerous drugs: 2 compounds / day • Alzheimer's / Parkinson efficacy assays: 2-6 compounds / day • Angiogenesis assay: Test of inhibition / activation of angiogenesis for regenerative or efficacy of anticancerous drugs: with 2-10 compounds per day - 170 - Directory of Screening Platforms and Chemical Libraries in Spain Anexos BIOBIDE S.L. - 171 - Directory of Screening Platforms and Chemical Libraries in Spain FUNDACIÓN GAIKER General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 172 - Directory of Screening Platforms and Chemical Libraries in Spain General Information FUNDACIÓN GAIKER Nombre y titularidad GAIKER FOUNDATION Inicio de actividades 1996 Contacto Nombre Maitane Ipiñazar / Blanca Suárez Dirección postal Parque Tecnológico, Edificio 202 48170 Zamudio, Bizkaia Correo electrónico [email protected] [email protected] Teléfono +34 94 600 23 23 - 173 - Directory of Screening Platforms and Chemical Libraries in Spain Installations FUNDACIÓN GAIKER Usable surface area 450 m2 Authorization from Nuclear Safety Authority - (if necessary) Other (specify) - - 174 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries FUNDACIÓN GAIKER Management of chemical libraries for assays Storage capacity for compounds (number) 60 Storage conditions (temperature, concentration of solvents,...) 4ºC, room temperature , -20ºC, -80ºC, Liquid N2 Minimum volume accepted 1-50 ųl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 175 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available FUNDACIÓN GAIKER Targets available Types of assay Luminiscence Colorimetric Fluorescence Surface Plasmon Resonance Flow cytometry HPLC/MS/MS Others: In vitro Toxicity, In vitro Metabolism, In vitro drug inhibition and interaction of CYP450, In vitro Permeability, Pharmacogenomics/Toxicogenomics, Pharmacoproteomics/Toxicoproteomics Assay format 96 well plates 384 well plates - 176 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents FUNDACIÓN GAIKER Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Protein production and purification Growth conditions Monolayer Production capacity for biological reagents (nº of experimental points/week) - - 177 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure FUNDACIÓN GAIKER Description (robotized systems, detectors, etc) • CELL CULTURE LABORATORY • RISK 3 LABORATORY • MICROBIOLOGY LABORATORY • MOLECULAR BIOLOGY LABORATORY • RNA AND GENOMICS LABORATORY • PROTEINS AND PROTEOMICS LABORATORY • INSTRUMENTAL ANALYSIS LABORATORY • ACQUITY UPLC (triple quadrupole) • AKTA EXPLORER • RNA-DNA ANALYSER (Agilent 2100 Bioanalyzer) • TANGENTIAL FILTRATION CELL OPTISEP • HPLC DAD LIQUID CHROMATOGRAPH • HPLC-MASS /DAD LIQUID CHROMATOGRAPH • DNA MICROARRAY SCANNER (Agilent G2565CA) • 2D ELECTROPHORESIS • 3, 5 and 40-LITRE FERMENTERS • FLUORESCENCE READER • GEL READER • ELISA MULTISCAN PLATE READER • FREEZE-DRIER • FLUORESCENCE MICROSCOPE • QIACUBE • MULTICOLOUR REAL-TIME THERMOCYCLER • SURFACE PLASMON RESONANCE • FLOW CYTOMETER - 178 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel FUNDACIÓN GAIKER Fixed contract Nº % Doctors 7 24.1 Postgraduates 5 4.1 Qualified (diploma level) - 17.2 Technicians 5 17.2 Temporary contract Function R&D projects management, Group leader R&D projects management, Group leader Nº % Function R&D projects management, Group leader R&D projects management, Group leader 4 13.7 2 3.4 - - - - Laboratory 3 10.3 Laboratory 3 10.3 Laboratory (own R&D projects) Other personnel (specify) - 179 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations FUNDACIÓN GAIKER Validation criteria per plate assayed Good Laboratory Practices Validation criteria per screening survey - Validation criteria concentration-response curves Good Laboratory Practices Software for analysis of results Genomics: GeneSpring GX software (Agilent), Ingenuity software (Ingenuity Systems). Proteomics: SameSpots v3 (Nonlinear Dynamics) Database for storage and consultation of results Accreditations and audits available Microsoft Access/ChemFinder • GLP Compliance Certificate, BPLI/07.11/006MSC, issued by the Ministry of Health and Consumption to carry out " in vitro Toxicity " studies on pharmaceutical products, according to 2004/9/CE Directive and Royal Decree 2043/1994. • Company Registration Certificate ER- 0986/1998, issued by AENOR, with scope for the “Research, Development and Transfer of Technologies related to Plastic Materials, Recycling and Recovery, Environment and Biotechnology”, in accordance with UNE-EN ISO 9001:2008 Standard. • Environmental Management Certificate GA-2003/0107, issued by AENOR, with scope for the “Research, Development and Transfer of Technologies related to Plastic Materials, Recycling and Recovery, Environment and Biotechnology”, in accordance with UNE-EN ISO 14001:2004 Standard. • Validation VDM-05/051(European EMAS - Eco-Management and Audit Scheme), issued by AENOR which certifies that our Environmental Management System for “Research, Development and Transfer of Technologies related to Plastic Materials, Recycling and Recovery, Environment and Biotechnology” and the information included in our Environmental Statement are in accordance with European Regulation 761/2001 of 19th March 2001. - 180 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience FUNDACIÓN GAIKER Year Research projects Contracts with industry (and size) Screening campaigns 2004 2005 2006 2007 2008 3 5 7 5 5 2 (mediumlarge) 2 (mediumlarge) 3 (mediumlarge) 3 (mediumlarge) 3 (mediumlarge) - - - - - 2 2 2 2 2 Maximum yield per day Assays developed/optimized - 181 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered FUNDACIÓN GAIKER As services • in vitro Toxicity: • Cytotoxicity: Inhibition of cell proliferation, cell viability, total protein, mitochondrial activity • Genetic toxicology: 1. Test for inverted mutation in S. typhimurium (Ames Test). OECD Guideline for testing of chemical nº 471 2. Test for genetic mutation in mammal cells. OECD Guideline for testing of chemicals nº 476 • in vitro Metabolism: • Definition of the metabolisation pattern • Kinetic metabolisation study: Km, Ke, Vmax • Development and completion of chromatographic technique • Characterisation of metabolites • Comparative study of metabolisation pattern in hepatic systems of different species • Identification of CYP450 isozymes • Monitoring phase I and phase II enzyme activity • Bioavailability • Permeability in vitro • Permeability coefficient calculation (Papp) • in vitro Cell activity • Protein binding • Haemolysis • Biological assessment of medical materials (ISO 10993) • Toxicogenomics/Pharmacogenomics • Toxicoproteomics/Pharmacoproteomics Our experimental systems include: 3T3, L-929, HepG2, MCF7, CHO, human cell line CACO-2, NCM-460, freeze-dried human hepatocytes, hepatic microsomes, erythrocytes, human chondrocytes, human keratinocytes - 182 - Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered FUNDACIÓN GAIKER For collaboration • Development and validation of new cell models of physiological barriers of interest in the field of the pharmaceutical and cosmetics industry, such as the blood-brain, corneal, skin and lung barriers. • Nanomedicine (Nanotoxicology, Nanopharmacology and activity) • Microencapsulation of drugs with supercritical and conventional fluid technology (interfacial polymerisation, coacervation and spraydrying) - 183 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes FUNDACIÓN GAIKER - 184 - Directory of Screening Platforms and Chemical Libraries in Spain INNOPROT S.L. General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 185 - Directory of Screening Platforms and Chemical Libraries in Spain General Information INNOPROT S.L. Name and ownership Innoprot SL (Innovative Technologies in Biological Systems SL) Start-up date September 2007 Contact information Name Clarisa Salado Postal address Innoprot SL Edificio 502. Planta 1- Parque Tecnológico de Bizkaia 48160 – Derio, Spain E-mail [email protected] Phone number +34 94 400 5355 - 186 - Directory of Screening Platforms and Chemical Libraries in Spain Installations INNOPROT S.L. Usable surface area 300 m2 Authorization from Nuclear Safety Authority (if necessary) NO Other (specify) - - 187 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries INNOPROT S.L. Management of chemical libraries for assays Storage capacity for compounds (number) 5000 Storage conditions (temperature, concentration of solvents,...) 4º, -20º and -80ºC Minimum volume accepted 1-50 µl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 188 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available INNOPROT S.L. Targets available GPCRs Ion channels Peptide receptors Kinases Receptors with enzymatic activity Other enzymes Transporters Types of assay Luminiscence Colorimetric Fluorescence Polarised fluorescence TRF FRET BRET Assay format 96-well plates 384-well plates - 189 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents INNOPROT S.L. Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Protein production and purification Growth conditions Monolayer Co-culture Production capacity for biological reagents (nº of experimental points/week) - - 190 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure INNOPROT S.L. Description (robotized systems, detectors, etc) • BDTM Pathway 855 Bioimager is a HCA/HCS systems with a switchable software, confocal imaging capability. Real-time viewing of confocal images through the built in binocular eyepiece and the rapid capture of single confocal digital images, or, z-stacks of confocal images in automated assay mode are all made possible by incorporation of Nipkow spinning disk technology. The Nipkow disk can be moved in and out of the light path under manual, or fully automatic control and with the disk out of the light path, the BD Pathway 855 Bioimager operates as an inverted wide-field epi-fluorescence microscope. If required, this capability extends to automatic switching between confocal and wide-field modes for alternate fluorescence channels within a High Content screening assay. • The Microlab STAR line workstation is based on superior air displacement pipetting technology. Our workstation is configured with arms, with a high precision 96 channel head, adaptable to a wide range of labware including 96, 384, and 1536 well microtiter plates arm the iSWAP arm, a gripper tool that can access items on or off the deck. This robotic gripper is highly flexible with its vertical rotary movement capabilities. Third-party devices, as the microscope and the microplate reader are integrated in the workstation controlled by the Venus software. • The Synergy 2 is BioTek's high-performance multi-mode plate reader designed for the life science research and drug discovery . The detection modes include Fluorescense Intensity, Time-Resolved Fluorescence, Fluorescence Polarization, Glow and Flash Luminescence, UV-Vis Absorbance, FRET, TR-FRET, BRET, Area Scanning, Spectral Scanning - 191 - two and and and Directory of Screening Platforms and Chemical Libraries in Spain Personnel INNOPROT S.L. Fixed contract Nº % Function Doctors 5 70 Project manager Postgraduates 1 50 R&D and services Qualified (diploma level) 1 50 Lab assistant Technicians 4 70 Other personnel (specify) - 192 - Temporary contract Nº % Function Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations INNOPROT S.L. Validation criteria per plate assayed Z factor, signal to noise ratio, controls Validation criteria per screening survey Z value distribution Validation criteria concentration-response curves IC50 of standards Software for analysis of results Attovision, BDdataexplorer, IPlab, biotek´Gen5data analysis, Spotfire, CellVis (proprietary software) Database for storage and consultation of results - Accreditations and audits available - - 193 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience INNOPROT S.L. Year 2004 Research projects Contracts with industry (and size) 2005 2006 2007 2008 (2009) 1 4 5 4 12 Screening campaigns 6 Maximum yield per day * Assays developed/optimized 15 - 194 - 35 Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered INNOPROT S.L. As services • Receptor activation: GPCR internalization, Transfluor™, Labeled ligand internalization, Calcium flux, Cell membrane receptor binding, Nuclear hormone receptors • Toxicity and apoptosis: Chromatin condensation, Cell proliferation, Nuclear count, Mitochondrial integrity, Annexin V binding, Viability/cell count, Cell rounding, Micronuclei • Cell cycle: Cell cycle status reporting, DNA replication studies, Cell proliferation, Muliplexed cell cycle analysis, Cell cycle (DNA content) • Signalling pathways: Transcription factor activation & deactivation, Cell survival signalling, Cell migration signalling, Stress response signalling, Reporter gene expression, Protein expression • Translocations: Nucleo-cytoplasmic translocation, Plasma membrane translocation, Mitochondrial translocation • Cell morphology: Endothelial cell tube formation (angiogenesis) and Neurite outgrowth (neurotoxicity) • Cell differentiation assays using human primary cells For collaboration • Receptor activation: GPCR internalization, Transfluor™, Labeled ligand internalization, Calcium flux, Cell membrane receptor binding, Nuclear hormone receptors • Toxicity and apoptosis: Chromatin condensation, Cell proliferation, Nuclear count, Mitochondrial integrity, Annexin V binding, Viability/cell count, Cell rounding, Micronuclei • Cell cycle: Cell cycle status reporting, DNA replication studies, Cell proliferation, Muliplexed cell cycle analysis, Cell cycle (DNA content) • Signalling pathways: Transcription factor activation & deactivation, Cell survival signalling, Cell migration signalling, Stress response signalling, Reporter gene expression, Protein expression • Translocations: Nucleo-cytoplasmic translocation, Plasma membrane translocation, Mitochondrial translocation • Cell morphology: Endothelial cell tube formation (angiogenesis) and Neurite outgrowth (neurotoxicity) • Cell differentiation assays using human primary cells - 195 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes INNOPROT S.L. - 196 - Directory of Screening Platforms and Chemical Libraries in Spain Pharmakine S.L. General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 197 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Pharmakine S.L. Name and ownership Dominion Pharmakine S.L. Start-up date 2002 Contact information Name Erika Sáenz Postal address Parque tecnológico de Bizkaia Edificio 801ª Derio, 48160 Bizkaia - Spain E-mail mailto:[email protected] Phone number +34 944317037 - 198 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Pharmakine S.L. Usable surface area 525 m2 Authorization from Nuclear Safety Authority (if necessary) - Other (specify) - - 199 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries Pharmakine S.L. Management of chemical libraries for assays Storage capacity for compounds (number) Storage conditions (temperature, concentration of solvents,...) Minimum volume accepted Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 200 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Pharmakine S.L. Targets available Peptide receptors Types of assay Luminiscence Colorimetric Fluorescence Flow cytometry Assay format 96-well plates 24-well plates 48-well plates - 201 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Pharmakine S.L. Does the organization produce its own biological reagents? YES The unit has the capacity for the following Primary cultures Growth conditions Monolayer Production capacity for biological reagents 5 points/week (nº of experimental points/week) - 202 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Pharmakine S.L. Description (robotized systems, detectors, etc) - Animal facilities comprising animal handling room (96 cages for mice and 24 cages for rats), cage wash-up room, SAS entry/exit system, pressurize rooms, control of humidity, Tª and light cycles - Two white rooms under ISO 7 according to UNE-EN ISO 14644-1 - Fx Pro Imaging System (Kodak) for both in vitro and in vivo tumor tracking at real time using fluorescence or luminescence, integrating also an X Ray approach. The system is fully automated and has a digital camera with CCD technology of 2048 x 2048 pixels and it is operated with a software from Kodak Molecular Imaging / Carestream Molecular Imaging. - Multiskan AFCENT, an ELISA reader compatible with 96- and 384-well plates. Its highly accuracy detects fluorescence at 340, 405, 450, 492, 540 and 620 nm. - BD FACSCalibur Flow Cytometer of double laser technology with capabilities for four different fluorescences and an integrated Data Analysis system. - Fluoroskan Ascent, a versatile florimeter compatible with 6- to 384-well plates, excitation filters of 355, 485 and 530 nm and emission filters of 460, 538 and 620 nm. - Optimax plus Biogenex Inmunostainning system with a capacity of 40 slides and a software that automates the control Tª and antibody amounts. - Tissue Arrayer for gathering up to 1000 different paraffin samples into one single slide - Agilent DNA microarray scanner Bundle (G2565BA) for gene expression profile analysis with array formats of 1X244K, 2X105K, 4X44K, 8X15K. It also determines gains and losses in the genome. - ABI PRISM® 7000 Sequence Detection System, a Real Time RT-PCR system to quantify nucleic acids. It works with 96-well plates. - 203 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel Pharmakine S.L. Fixed contract Nº % Function Doctors 6 70 Responsible for project or service. R+D Management. In charge of Technology Units. Postgraduates 4 30 Administration. Quality. Commercial management. Technicians 5 40 Laboratory technitian Other personnel (specify) 1 40 Laboratory assistant Temporary contract Nº Qualified (diploma level) - 204 - % Function Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Pharmakine S.L. Validation criteria per plate assayed Positive and negative internal controls for each assay. Validation criteria per screening survey - Validation criteria concentration-response curves - Software for analysis of results - Database for storage and consultation of results - Accreditations and audits available Internal auditories carried out by Quality Department. GLP procedures. - 205 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience Pharmakine S.L. Year 2004 2005 2006 2007 2008 (2009) Research projects 2 3 4 4 4 4 Contracts with industry (and size) 1 1 2 2 3 3 Screening campaigns 0 0 0 0 0 0 Maximum yield per day 0 0 0 0 0 0 Assays developed/optimized 2 2 6 8 20 20 - 206 - Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Pharmakine S.L. As services - Citotoxicity assays on primary isolated murine cells including hepatocytes, fibroblasts, astrocytes, renal cells, tumor cells... 30 compounds/plate in 96-well plate format. Ten plates/day - Tumor cell adhesion assays on primary cells (monolayer), 30 compounds/plate, 96-well plate format and 24-well plates (6 compounds per plate). - 3D cell culture proliferation assays, 30 compounds/plate in 96-well plate format. Ten plates/day - In vivo murine models for anti-cancer efficacy studies. Xenografts, orthotopic and metastatic experimental assays. For collaboration - Citotoxicity assays on primary isolated murine cells including hepatocytes, fibroblasts, astrocytes, renal cells, tumor cells... 30 compounds/plate in 96-well plate format. Ten plates/day - Tumor cell adhesion assays on primary cells (monolayer), 30 compounds/plate, 96-well plate format and 24-well plates (6 compounds per plate). - 3D cell culture proliferation assays, 30 compounds/plate in 96-well plate format. Ten plates/day - In vivo murine models for anti-cancer efficacy studies. Xenografts, orthotopic and metastatic experimental assays. - 207 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Pharmakine S.L. - 208 - Directory of Screening Platforms and Chemical Libraries in Spain Biopolis S.L. Translation is still pending General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 209 - Directory of Screening Platforms and Chemical Libraries in Spain Datos de adscripción y titularidad Name and ownership BIOPOLIS, S.L. Start-up date 23 de abril de 2003 Biopolis S.L. Contact information Name Nina Coolsaet, Patricia Martorell Postal address Parc Científic Universitat de València, Edificio 2, Biotech C/ Catedrático Agustín Escardino Benlloch, 9, 46980 Paterna, Valencia, España E-mail [email protected], [email protected] Phone number +34963160299 - 210 - Directory of Screening Platforms and Chemical Libraries in Spain Instalaciones Biopolis S.L. Usable surface area 1.500 m2 Authorization from Nuclear Safety Authority (if necessary) No se requiere Other (specify) Autorización para trabajar con organismos genéticamente modificados - 211 - Directory of Screening Platforms and Chemical Libraries in Spain Gestión de quimiotecas Biopolis S.L. Management of chemical libraries for assays Storage capacity for compounds (number) Storage conditions (temperature, concentration of solvents,...) Minimum volume accepted Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 212 - Directory of Screening Platforms and Chemical Libraries in Spain Ensayos disponibles Biopolis S.L. Targets available Otras enzimas: Lipasa pancreática (LP) Prolil endopeptidasa (PEP) Enzima convertidora de Angiotensina (ACE) Fosfolipasa A2 α-Ramnosidasa β-glucosidasa α-arabinofuranosidasa Types of assay Colorimétricos Fluorescencia Quimiluminiscencia HPLC/MS/MS GC/MS Otros Modelos propios de evaluación in vivo Saccharomyces cerevisiae Caenorhabditis elegans Pez Medaka Assay format 12-pocillos 96-pocillos 386-pocillos - 213 - Directory of Screening Platforms and Chemical Libraries in Spain Producción de reactivo biológico Biopolis S.L. Does the organization produce its own biological reagents? YES The unit has the capacity for the following Técnicas de biología molecular: clonaje, mutagenesis, análisis de expresión. Producción de compuestos bioquímicos en sistemas bacterianos, levaduras, hongos filamentosos. Producción, purificación y caracterización de proteínas recombinantes. Biosíntesis, purificación y análisis de compuestos orgánicos. Desarrollo de modelos rápidos de escrutinio Growth conditions Monocapa Spinner flasks Biorreactores Production capacity for biological reagents (nº of experimental points/week) Variable - 214 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructura Biopolis S.L. Description (robotized systems, detectors, etc) • Para screening en S. cerevisiae: equipos de incubación/lectura continua del crecimiento microbiano para placas de 96 pocillos (Multiscan). • Para screening en C. elegans: sistema de monitorización automático compatible con placas de 96/386 pocillos. • Microrreactor automático (24μ-bioreactor/Applikon) para 24 bioreacciones simultáneas en 3-12 mL en condiciones controladas e independientes de pH, oxígeno y temperatura. • Unidad de cultivos celulares • Termocicladores de gradiente y de tiempo real. • Sistemas de visualización y documentación de imágenes para geles, membranas y placas de 96 pocillos para luminiscencia, fluorescencia, y quimiluminiscencia. • Espectrofotómetros de barrido UV/Vis. • Espectrofluorímetro de barrido. • Equipos de cromatografía líquida de alta presión (HPLC), analíticos y semipreparativos, con detectores acoplados de fotodiodo, índice de refracción, luz dispersiva (Waters) • Equipos de cromatografía líquida de baja presión, con detector de ultravioleta-visible (Äkta Explorer/Äkta Pilot Chromatography system, GE-Healthcare) para la optimización y purificación preparativa de compuestos bioquímicos. • Equipo de Cromatografía de Gases (Agilent). - 215 - Directory of Screening Platforms and Chemical Libraries in Spain Personal Biopolis S.L. Fijo Nº % Función Doctors 14 43 Postgraduates 9 27 Qualified (diploma level) 6 18 Técnicos del laboratorio Technicians 4 12 Administración Contratado Nº Investigadores/Gestión de proyectos Investigadores/Gestión de la empresa Other personnel (specify) - 216 - % Función Directory of Screening Platforms and Chemical Libraries in Spain Control de calidad y acreditaciones Biopolis S.L. Validation criteria per plate assayed N/A Validation criteria per screening survey N/A Validation criteria concentration-response curves N/A Software for analysis of results N/A Database for storage and consultation of results N/A Accreditations and audits available En desarrollo: ISO 9001 / GMP / GLP - 217 - Directory of Screening Platforms and Chemical Libraries in Spain Experiencia en Screening Biopolis S.L. Año 2004 2005 Research projects 2006 2007 2008 2 2 5 2 2 4 (150000€) (180000€) (200000€) Contracts with industry (and size) Screening campaigns Maximum yield per day Assays developed/optimized 3 - 218 - 7 8 (2009) Directory of Screening Platforms and Chemical Libraries in Spain Capacidades disponibles para ofertar Biopolis S.L. Como servicio Los ensayos descritos en continuación consisten en ensayos preclínicos in vivo con el modelo animal C. elegans (1-7), el modelo eucariota S. cerevisiae (8-10) y los ensayos in vitro (11-14). 1) Ensayo “life span” en C. elegans Tipo de ensayo: Manual (placas). Duración ensayo: 3 semanas Capacidad máxima: 10 compuestos/3 semanas 2) Ensayo de estrés oxidativo en C. elegans Tipo de ensayo: placa. Duración ensayo: 7 días Capacidad máxima: 12 compuestos/7 días Tipo de ensayo: placa microtitulación 12 pocillos. Duración ensayo: 1 semana Capacidad máxima: aprox. 50 compuestos/1 mes 3) Ensayo de obesidad por fluorimetría en C. elegans Tipo de ensayo: medida fluorescencia manual. Duración ensayo: 7 días Capacidad máxima: 12 compuestos/7 día 4) Ensayo análisis composición de lípidos en C. elegans Tipo de ensayo: cuantificación triglicéridos y fosfolípidos (kits y ensayo colorimétrico): Capacidad máxima: 12 compuestos/7 día Tipo de ensayo: cuantificación por cromatografía 5) Ensayo de Alzheimer en C. elegans (reducción de parálisis) Tipo de ensayo: manual (placa). Duración del ensayo: 2 semanas Capacidad máxima: 10 compuestos/2 semanas 6) Ensayo de toxicidad de compuestos en C. Elegans Tipo de ensayo: manual (placa). Duración del ensayo: 3 semanas Capacidad máxima: 10 compuestos/3 semanas 7) Ensayo de dermoprotección de compuestos en C. Elegans Tipo de ensayo: manual (placa). Duración del ensayo: 2 semanas Capacidad máxima: 10 compuestos/2 semanas - 219 - Directory of Screening Platforms and Chemical Libraries in Spain Capacidades disponibles para ofertar (cont.) Biopolis S.L. Como servicio (continúa) 8) Ensayo de estrés oxidativo en S. cerevisiae Tipo de ensayo: Placa multipocillo (96) en lector/incubador. Tiempo ejecución: 2 días Capacidad máxima: 96 compuestos/ 2 días 9) Ensayo de obesidad por fluorimetría en S. cerevisiae Tipo de ensayo: manual (fluorescencia). Duración: 2- 3días Capacidad máxima: 40- compuestos/2 días 10) Ensayo análisis composición de lípidos en S. cerevisiae por cromatografía Tipo de ensayo: manual (fluorescencia). Duración: 2- 3días Capacidad máxima: 10- compuestos/2 días 11) Ensayo in vitro de actividades enzimáticas relacionadas con obesidad, Alzheimer y celiaquía: Lipasa pancreática (LP), Prolil endopeptidasa (PEP), Enzima convertidora de Angiotensina (ACE) Tipo de ensayo: espectrofotometría. Duración del ensayo: Capacidad máxima: 20 compuestos /día Fosfolipasa A2 Tipo de ensayo: Cromatografía Capacidad máxima: 10 compuestos /día 12) Medida in vitro de actividades enzimáticas relacionadas con liberación de aromas: α-Ramnosidasa, β-glucosidasa, α-arabinofuranosidasa Tipo de ensayo: Placa multipocillo (96) en lector/incubador Capacidad máxima: 80 compuestos / día 13) Medida de la degradación del péptido 33-mer por cromatografía (HPLC) Capacidad máxima: 10 compuestos /día 14) Modelo digestión in vitro Capacidad máxima: 10 compuestos /día Como colaboración: Mismos que cómo servicio - 220 - Directory of Screening Platforms and Chemical Libraries in Spain Anexos Biopolis S.L. - 221 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 222 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma General Information Name and ownership In vivo Sreening Platform in Drosophila melongaster models, VALENTIA BioPharma Start-up date 2006 Contact information Name Rebeca Lucas M. Carmen Álvarez Postal address VALENTIA BioPharma SL Parc Cientific Universitat Valencia; Pol La Coma s/n 46980 Paterna, Valencia E-mail [email protected] [email protected] Phone number +34 963 543 842 - 223 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Installations Usable surface area 100 m2 Authorization from Nuclear Safety Authority (if necessary) University of Valencia Building Other (specify) - - 224 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Management of chemical libraries Management of chemical libraries for assays Storage capacity for compounds (number) 100.000 in 96-well plate Storage conditions (temperature, concentration of solvents,...) -20ºC and room temperature, DMSO, other Minimum volume accepted From 1 µl to 500 µl Management of own chemical libraries Does the organization have its own chemical library? NO Format of the chemical library Coding method used in the chemical library State of compounds in the chemical library Number of compounds Mean molecular weight of the compounds (include standard deviation) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Degree of purity of compounds in the chemical library (mean value) Exclusion criterion for purity (% cut-off) - 225 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Plataforma de Screening in vivo en Drosophila melanogaster, VALENTIA BioPharma Targets available Molecular biology iRNA Types of assay Luminiscence Colorimetric Fluorescence Polarized fluorescence FRET (partner compamy) Flow cytometry (cellular aggregates) In vivo screening in Drosophila melanogaster Assay format 96-well plates Vals of different sizes placed in 96-positions plate - 226 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Production of biological reagents Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Growth conditions Automatic dosage of D.melanogaster and C.Elegans embryos Production capacity for biological reagents (nº of experimental points/week) - - 227 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Description (robotized systems, detectors, etc) - Union Biometrica; Copas Select EmbryoSorter large particles flow cytometer - Perkin-Elmer; Envision Xcite multireader with stackers - Beckman; Biomek NXp 96 head Liquid handler with stackers - Beckman; Biomek NXp 8 head Liquid handler with stackers (2Q2010) - 228 - Directory of Screening Platforms and Chemical Libraries in Spain Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Personnel Fixed contract Nº % Function Temporary contract Nº % Function Doctors 1 100 Scientific Director 1 50 R+D Director Postgraduates 2 100 Management 1 50 R+D Qualified (diploma level) 2 100 R+D Technicians 1 100 Auxiliar Lab 2 100 Advisor Other personnel (specify) - 229 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Validation criteria per plate assayed Z factor, Z score, SD, CV, QC, edge effect, inflexion point Validation criteria per screening survey Z’ factor, internal control, screening window Validation criteria concentration-response curves IC50, internal control, screening window Software for analysis of results JMP 7, own developing Database for storage and consultation of results - Accreditations and audits available Good laboratory practice- OCDE in process of implementation - 230 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma Year 2004 2006 2007 2008 (2009) 2 4 4 (6) Contracts with industry (and size) 1 2 (3) Screening campaigns 1 1 (6) Maximum yield per day 24 80 (480) 2 (6) Research projects Assays developed/optimized - 231 - 2005 Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma As services - Screening of drugs in two in vivo D. melanogaster models of Myotonic Dystrophy (phenotypic models) - Screening of drugs in four in vivo D. melanogaster models of Myotonic Dystrophy (biochemical models) - Screening of anti-inflammatory and antioxidant drugs in in vivo D. melanogaster models - Capacity 160x3:480 wells/day/assay (assay duration 14 days) For collaboration - Development of new Drosophila melanogaster models of genetic diseases - Longevity assay in Drosophila models - In vivo mutagenesis assay in Drosophila models - 232 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Plataforma de Screening in vivo en Drosophila melanogaster , VALENTIA BioPharma - 233 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Investigación Príncipe Felipe General Information Installations Management of chemical libraries Assays available Production of biological reagents Description of infrastructure Personnel Quality control and accreditations Screening experience Available capacity offered Annexes - 234 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Fundación Centro de Investigación Príncipe Felipe Name and ownership Fundación Centro de Investigación Príncipe Felipe Start-up date 2005 Contact information Name María Jesús Vicent Docón Postal address Av. Autopista del Saler 16 46012 Valencia E-mail [email protected] Phone number +34 963289680 - 235 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Fundación Centro de Investigación Príncipe Felipe 150 m2 Usable surface area Authorization from Nuclear Safety Authority (if necessary) YES Other (specify) Laboratory surface area, RMN area, molecular biology area, -80ºC room, cell culture room - 236 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries Fundación Centro de Investigación Príncipe Felipe Management of chemical libraries for assays Storage capacity for compounds (number) 14.000 cp Storage conditions (temperature, concentration of solvents,...) -80ºC, 5mM, DMSO Minimum volume accepted 1-50ųl Management of own chemical libraries Does the organization have its own chemical library? YES, three libraries: MYRIASCREEN, PRESTWICK, CEREP Format of the chemical library 96-well plates Coding method used in the chemical library Alphanumerical codes State of compounds in the chemical library Solid, Solution (MYRIASCREEN and PRESTWICK in DMSO, 5mM concentration) Number of compounds MYRIASCREEN 10000; PRESTWICK 1120; CEREP 686 Mean molecular weight of the compounds (include standard deviation) MYRIASCREEN 328.1; PRESTWICK 385.8; CEREP 220.6 Characteristics of the chemical library Average number of H donors and acceptors MYRIASCREEN 3.0 and 1.3; PRESTWICK 4.8 and 2.9; CEREP 2.6 and 1.5 Percentage of natural products 0,00% Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -80 ºC on dark Degree of purity of compounds in the chemical library (mean value) CEREP 99.6%; PRESTWICK 98%; MYRIASCREEN 98% Exclusion criterion for purity (% cut-off) <98% - 237 - Directory of Screening Platforms and Chemical Libraries in Spain Assays available Fundación Centro de Investigación Príncipe Felipe Targets available Ion channels Peptide receptors Receptors with enzymatic activity Kinases Trascription factor HRE, CRE and TRE PAG (Phosphate activated Glutaminase) Types of assay Radiactive Luminiscence Colorimetric Fluorescence Polarised fluorescence Flow cytometry HPLC/MS/MS Zebra fish Others: - Assay format Screen Validation. (in vitro, cell-based & embryo-based screening medaka fish). Statistical validation; Q-analysis. dB –Management. MedChem interaction. HTP-Screening. Secondary evaluation of hits; hit to Lead processes. Information Management. Support for intellectual property. Animal models; identification and validation. 96-well plates - 238 - Directory of Screening Platforms and Chemical Libraries in Spain Production of biological reagents Fundación Centro de Investigación Príncipe Felipe Does the organization produce its own biological reagents? YES The unit has the capacity for the following Cloning Cell line expression Primary cultures Protein production and purification Growth conditions Monolayer Suspensión cell culture Production capacity for biological reagents (nº of experimental points/week) 5.000 - 239 - Directory of Screening Platforms and Chemical Libraries in Spain Infraestructure Fundación Centro de Investigación Príncipe Felipe Description (robotized systems, detectors, etc) - Robot Freedom Evo (Tecan). TE-MO 96 Multi-channel pipetting option. Victor3 (Fluorescence, Luminiscence, Absorbance). Victor 2v (Polarized Fluorescence, Fluorescence Anisotropy) Common Equipment Full-equipped Tissue culture room (A family of cell lines available: cancer, primary and stem cells) - 240 - Directory of Screening Platforms and Chemical Libraries in Spain Personnel Fundación Centro de Investigación Príncipe Felipe Fixed contract Nº % Temporary contract Function Doctors Nº % Function 2 50 Service responsible Experimental design Validation criteria Secondary evaluation of hits; hit to Lead processes 1 100 Equipment set up Experimental lab-work Postgraduates Qualified (diploma level) Technicians Other personnel (specify) - 241 - Directory of Screening Platforms and Chemical Libraries in Spain Quality control and accreditations Fundación Centro de Investigación Príncipe Felipe Validation criteria per plate assayed Reference inhibitors Assay 1 compound: 1 well (80 compounds/plate) Positive validation: 1 compound: 4 wells Validation criteria per screening survey Activity (IC50), Selectivity/specificty, Toxicity Validation criteria concentration-response curves 4serial concentrations by compound (N=3 experiments) Software for analysis of results DIANA Software designed in the CIPF CIPF for stadistical data treatment and hit selection based in this equation: Z’= 1- [3xSDT+3xSDB] |XT-XB| T and B are the controls of the assay; T= total reaction ; B = background reaction Database for storage and consultation of results EXCELL CHEMFILE (MW, chemical structure, storage localization) DIANA SPF (CEREP library) Accreditations and audits available NO - 242 - Directory of Screening Platforms and Chemical Libraries in Spain Screening experience Fundación Centro de Investigación Príncipe Felipe Year 2004 2005 2006 2007 2008 2 4 6 6 3 3 1 1 2 1000 1000 1000 4 5 6 Research projects Contracts with industry (and size) Screening campaigns Maximum yield per day Assays developed/optimized 3 - 243 - (2009) Directory of Screening Platforms and Chemical Libraries in Spain Available capacity offered Fundación Centro de Investigación Príncipe Felipe As services • Compound Collection. • Screen Validation. (in vitro, cell-based & embryo-based screening medaka fish). • Statistical validation; Q-analysis. • dB –Management. • MedChem interaction. • HTP-Screening. • Secondary evaluation of hits; hit to Lead processes. • Information Management. • Support for intellectual property. • Animal models; identification and validation. For collaboration Same as above - 244 - Directory of Screening Platforms and Chemical Libraries in Spain Annexes Fundación Centro de Investigación Príncipe Felipe - 245 - Directory of Screening Platforms and Chemical Libraries in Spain Quimiotecas Biomar; Instituto Biomar, S.A. Translation is still pending General Information Installations Management of chemical libraries for assays Management of own chemical libraries Biological/pharmacological annotation of the chemical library - 246 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Quimiotecas Biomar; Instituto Biomar, S.A. Name and ownership Quimiotecas Biomar; Instituto Biomar, S.A. Start-up date 1996 Contact information Name Jesús Ángel de la Fuente Postal address Instituto Biomar, S.A Parque Tecnológico de León, Parcela M-10.4 24009 Armunia (León), Spain E-mail [email protected] Phone number +34 91 8441807 - 247 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Quimiotecas Biomar; Instituto Biomar, S.A. Usable surface area 2.300 m2 Authorization from Nuclear Safety Authority (if necessary) - Other (specify) - - 248 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries for assays Quimiotecas Biomar; Instituto Biomar, S.A. Storage capacity for compounds (number) - Storage conditions (temperature, concentration of solvents,...) -20ºC, secos/DMSO Minimum volume accepted 1-50 μl - 249 - Directory of Screening Platforms and Chemical Libraries in Spain Management of own chemical libraries Quimiotecas Biomar; Instituto Biomar, S.A. Format of the chemical library Tubos, placas de 96 pocillos Coding method used in the chemical library Códigos alfanuméricos State of compounds in the chemical library Sólido Number of compounds En la actualidad existen tres quimiotecas diferentes: BNPL-E: con 40.000 extractos naturales de microorganismos BNPL-S: con 1.500 compuestos naturales aislados de microorganismos y sin estructura química asignada BNPL-C: con 500 compuestos naturales aislados de micoorganismos y con estructura química asignada Mean molecular weight of the compounds (include standard deviation) 487±271 (para la quimioteca BNPL-C) Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products prácticamente el 100% Does the library contain mixtures of natural products? YES Storage conditions (temperature, atmosphere, light) - Degree of purity of compounds in the chemical library (mean value) >80% (HPLC) para las quimiotecas BNPL S y C Exclusion criterion for purity (% cut-off) - Software for management of the chemical library Access, ChemFinder Maximum number of freeze/thawing cycles permitted in the chemical library Origin of compounds Propios Intellectual property of the chemical library - Does the unit have directed sub-libraries? NO - 250 - - Directory of Screening Platforms and Chemical Libraries in Spain Biological/pharmacological annotation of the chemical library Quimiotecas Biomar; Instituto Biomar, S.A. Is the chemical library annotated? YES How is the chemical library annotated? Experimental annotation Description of the annotation Citotoxicity Actividad antibacteriana Actividad Antifúngica. - 251 - Directory of Screening Platforms and Chemical Libraries in Spain Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) General Information Installations Management of chemical libraries for assays Management of own chemical libraries Biological/pharmacological annotation of the chemical library - 252 - Directory of Screening Platforms and Chemical Libraries in Spain Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) General Information Name and ownership Chembiobank – Drug Discovery Platform – Barcelona Science Park (Parc Científic Barcelona (PCB)) Start-up date 2007 Contact information Name Jordi Quintana Postal address C/ Baldiri Reixac, 10 08028 Barcelona, Spain E-mail [email protected] Phone number +34 93 4034557 / 636 981 982 - 253 - Directory of Screening Platforms and Chemical Libraries in Spain Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) Installations Usable surface area 50 m2 Authorization from Nuclear Safety Authority (if necessary) - Other (specify) - - 254 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries for assays Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) Storage capacity for compounds (number) 50.000 Storage conditions (temperature, concentration of solvents,...) From RT to -20ºC, 10mM DMSO Minimum volume accepted 1-50 µl - 255 - Directory of Screening Platforms and Chemical Libraries in Spain Management of own chemical libraries Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) Format of the chemical library Tubes; 96-well plates Coding method used in the chemical library Bar codes State of compounds in the chemical library Solid; Solution 10mM DMSO Number of compounds 50.000 (in construction) Mean molecular weight of the compounds (include standard deviation) 300 - 1000 Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -20ºC for compounds solutions Degree of purity of compounds in the chemical library (mean value) >95% (commercial), >90% (academic) Exclusion criterion for purity (% cut-off) >90% Software for management of the chemical library Activity Base (IDBS) (chemistry & biology), Mosaic (Titian) (logistics) Maximum number of freeze/thawing cycles permitted in the chemical library 2 Origin of compounds Commercial; Collaborations Intellectual property of the chemical library Published; Commercial compounds Does the unit have directed sub-libraries? NO - 256 - Directory of Screening Platforms and Chemical Libraries in Spain Biological/pharmacological annotation of the chemical library Chembiobank - Plataforma Drug Discovery - Parc Científic Barcelona (PCB) Is the chemical library annotated? NO, (in process) How is the chemical library annotated? Virtual annotation (in process) Experimental annotation (in process) Description of the annotation The goal of Chembiobank is to annotate the Chemical Library both virtually and experimentally through collaborations with entities in Spain and in Europe who have computational and HTS experimental screening assays available. - 257 - Directory of Screening Platforms and Chemical Libraries in Spain NEUROLAB_IQM General Information Installations Management of chemical libraries for assays Management of own chemical libraries Biological/pharmacological annotation of the chemical library - 258 - Directory of Screening Platforms and Chemical Libraries in Spain General Information NEUROLAB_IQM Name and ownership NEUROLAB_IQM (Instituto de Química Médica CSIC) Start-up date 1996 Contact information Name Dra. Ana Martínez Gil Postal address Instituto de Química Médica (CSIC) C/Juan de la Cierva 3 28006 Madrid E-mail [email protected] Phone number +34 91 5680010 - 259 - Directory of Screening Platforms and Chemical Libraries in Spain Installations NEUROLAB_IQM Usable surface area 5 m2 Authorization from Nuclear Safety Authority (if necessary) Not necessary Other (specify) - - 260 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries for assays NEUROLAB_IQM Storage capacity for compounds (number) > 2000 Storage conditions (temperature, concentration of solvents,...) Solid, room temperature, tubes Minimum volume accepted - - 261 - Directory of Screening Platforms and Chemical Libraries in Spain Management of own chemical libraries NEUROLAB_IQM Format of the chemical library Tubes Coding method used in the chemical library Alphanumerical codes State of compounds in the chemical library Solid Number of compounds >1000 Mean molecular weight of the compounds (include standard deviation) 200-500 Characteristics of the chemical library Average number of H donors and acceptors Percentage of natural products 0% Does the library contain mixtures of natural products? Storage conditions (temperature, atmosphere, light) Room temperature, darkness Degree of purity of compounds in the chemical library (mean value) 98% Exclusion criterion for purity (% cut-off) >95% Software for management of the chemical library FileMaker ChemFinder Maximum number of freeze/thawing cycles permitted in the chemical library - Origin of compounds Own organic synthesis Intellectual property of the chemical library Published obtention method Does the unit have directed sub-libraries? NO - 262 - Directory of Screening Platforms and Chemical Libraries in Spain Biological/pharmacological annotation of the chemical library NEUROLAB_IQM Is the chemical library annotated? NO How is the chemical library annotated? Description of the annotation - 263 - Directory of Screening Platforms and Chemical Libraries in Spain Fundación Centro de Investigación Príncipe Felipe General Information Installations Management of chemical libraries for assays Management of own chemical libraries Biological/pharmacological annotation of the chemical library - 264 - Directory of Screening Platforms and Chemical Libraries in Spain General Information Fundación Centro de Investigación Príncipe Felipe Name and ownership Fundación Centro de Investigación Príncipe Felipe Start-up date 2005 Contact information Name María Jesús Vicent Docón Postal address Av. Autopista del Saler 16 46012 Valencia E-mail [email protected] Phone number +34 963289680 - 265 - Directory of Screening Platforms and Chemical Libraries in Spain Installations Fundación Centro de Investigación Príncipe Felipe Usable surface area 150 m2 Authorization from Nuclear Safety Authority (if necessary) YES Other (specify) Laboratory surface area, RMN area, molecular biology area, -80ºC room, cell culture room - 266 - Directory of Screening Platforms and Chemical Libraries in Spain Management of chemical libraries for assays Fundación Centro de Investigación Príncipe Felipe Storage capacity for compounds (number) 14.000 cp Storage conditions (temperature, concentration of solvents,...) -80ºC, 5mM, DMSO Minimum volume accepted 1-50ųl - 267 - Directory of Screening Platforms and Chemical Libraries in Spain Management of own chemical libraries Fundación Centro de Investigación Príncipe Felipe There are 3 Chemical libraries: MYRIASCREEN 125 plates 96wells; PRESTWICK 14 plates 96wells; PCEREP Fragment based 7plates 96wells Format of the chemical library 96-well plates Coding method used in the chemical library Alphanumerical codes State of compounds in the chemical library Solid, Solution (MYRIASCREEN and PRESTWICK in DMSO, 5mM concentration) Number of compounds MYRIASCREEN 10000; PRESTWICK 1120; CEREP 686 Mean molecular weight of the compounds (include standard deviation) MYRIASCREEN 328.1; PRESTWICK 385.8; CEREP 220.6 Characteristics of the chemical library Average number of H donors and acceptors MYRIASCREEN 3.0 and 1.3; PRESTWICK 4.8 and 2.9; CEREP 2.6 and 1.5 Percentage of natural products 0,00% Does the library contain mixtures of natural products? NO Storage conditions (temperature, atmosphere, light) -80 ºC on dark Degree of purity of compounds in the chemical library (mean value) CEREP 99.6%; PRESTWICK 98%; MYRIASCREEN 98% Exclusion criterion for purity (% cut-off) <98% Software for management of the chemical library EXCELL; CHEMFILE; DIANA; SPF Maximum number of freeze/thawing cycles permitted in the chemical library 5 - 268 - Directory of Screening Platforms and Chemical Libraries in Spain Management of own chemical libraries Fundación Centro de Investigación Príncipe Felipe Origin of compounds Own Commercial Collaborations Intellectual property of the chemical library Commercial compounds Does the unit have directed sub-libraries? YES, Sintesized precursors or derivatized compounds - 269 - Directory of Screening Platforms and Chemical Libraries in Spain Biological/pharmacological annotation of the chemical library Fundación Centro de Investigación Príncipe Felipe Is the chemical library annotated? YES How is the chemical library annotated? Virtual annotation Experimental annotation Description of the annotation Biological activity - 270 -